Tesofensine 500mcg (30 Capsules)
$250.00
Tesofensine is a synthetic small-molecule research compound classified as a triple monoamine reuptake inhibitor of serotonin, norepinephrine, and dopamine. It modulates central appetite and metabolic signaling through presynaptic transporter inhibition and hypothalamic regulation in experimental models. Tesofensine is used in neuroendocrine and metabolic research investigating neurotransmitter dynamics, energy expenditure, and hypothalamic control mechanisms.
References:
Appel L et al., Neuropsychopharmacology, 2011 36(3):654–662
Astrup A et al., Lancet, 2008 372(9653):1906–1913
Eisenstein SA et al., J Pharmacol Exp Ther, 2015 352(1):129–138
These capsules are provided exclusively as inert containers for the dry transport, storage, and handling of laboratory research materials. They are not dosage forms and are not intended for ingestion or administration. They must not be used as drug delivery systems, consumed, or administered in any form. For Research Use Only: Not for use in humans or animals. Not for diagnostic or therapeutic purposes.
Tesofensine 500mcg (30 Capsules) – Buy High-Quality Tesofensine 500mcg (30 Capsules) Online
Looking to buy Tesofensine 500mcg (30 Capsules) for your research laboratory? You have come to the right place.
We currently have Tesofensine 500mcg (30 Capsules) for sale and it is in stock and ready for immediate shipping.
Our Tesofensine 500mcg (30 Capsules) is of the highest purity, making it the best Tesofensine 500mcg (30 Capsules) online for scientific studies.
Product Overview
Tesofensine 500mcg (30 Capsules) is a premium research compound widely utilized in various scientific studies.
Researchers seeking to buy Tesofensine 500mcg (30 Capsules) online often prioritize purity and consistency.
This compound has been studied extensively for its unique biochemical properties and its role in cellular pathways.
Overview
Tesofensine (NS-2330) is a synthetic small-molecule compound that has been described in the scientific literature within the context of monoamine transporter–associated research. Its characterization is based on biochemical, pharmacological, and observational datasets generated in controlled experimental and preclinical research environments. No therapeutic, diagnostic, or clinical claims are made or implied.
Biochemical Characteristics
Molecular Formula: C17H23Cl2NO
Molecular Weight: 327.27 g/mol
PubChem CID: 11370864
CAS Number: 195875-84-4
Alternative Names: Tesofensine, TE, NS-2330
Tesofensine is not a peptide and is structurally classified as a phenyltropane-derived compound. Published sources describe its use as a research compound in studies evaluating monoamine transporter–related biochemical parameters.
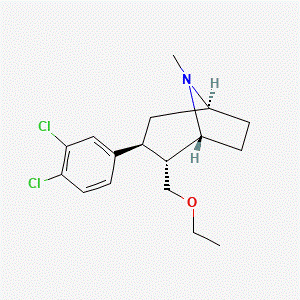
Source: PubChem
Research Applications
In research settings, tesofensine has been utilized as an experimental variable in:
- Monoamine transporter binding and uptake assays
- Neurochemical concentration measurements in animal models
- Behavioral and metabolic readout collection under controlled conditions
- Comparative pharmacology studies involving monoamine-associated compounds
All reported applications are limited to laboratory and preclinical research use.
Pathway / Mechanistic Context
Literature describing tesofensine references observed associations with monoaminergic pathway annotations derived from transporter occupancy data, neurotransmitter concentration profiles, and downstream signaling-related datasets. These descriptions are framed as reported experimental observations rather than asserted biological effects.
- Monoamine transporter–annotated datasets (dopaminergic, noradrenergic, serotonergic)
- Cholinergic pathway–adjacent measurements reported in selected models
- BDNF-annotated gene-expression and protein-level datasets
Preclinical Research Summary
Preclinical investigations involving tesofensine include rodent-based models in which researchers reported measured endpoints related to neurotransmitter dynamics, behavioral metrics, and metabolic parameters. Transcriptomic and proteomic analyses describe differential expression patterns among neurotrophic and metabolic gene sets under experimental exposure conditions.
Safety-related observations in the literature are reported as trial-level or study-level findings without extrapolation beyond the original experimental context.
Form & Analytical Testing
This research material is supplied with analytical documentation supporting identity and purity. Standard analytical techniques may include chromatography and mass spectrometry.
Referenced Citations
- K. A. Schoedel, D. Meier, B. Chakraborty, P. M. Manniche, and E. M. Sellers, “Subjective and objective effects of the novel triple reuptake inhibitor tesofensine in recreational stimulant users,” Clin. Pharmacol. Ther., vol. 88, no. 1, pp. 69–78, Jul. 2010, doi: 10.1038/clpt.2010.67.
- A. Astrup, D. H. Meier, B. O. Mikkelsen, J. S. Villumsen, and T. M. Larsen, “Weight loss produced by tesofensine in patients with Parkinson’s or Alzheimer’s disease,” Obes. Silver Spring Md, vol. 16, no. 6, pp. 1363–1369, Jun. 2008, doi: 10.1038/oby.2008.56.
- M. H. Larsen, H. Rosenbrock, F. Sams-Dodd, and J. D. Mikkelsen, “Expression of brain derived neurotrophic factor, activity-regulated cytoskeleton protein mRNA, and enhancement of adult hippocampal neurogenesis in rats after sub-chronic and chronic treatment with the triple monoamine re-uptake inhibitor tesofensine,” Eur. J. Pharmacol., vol. 555, no. 2–3, pp. 115–121, Jan. 2007, doi: 10.1016/j.ejphar.2006.10.029.
- A. Astrup, S. Madsbad, L. Breum, T. J. Jensen, J. P. Kroustrup, and T. M. Larsen, “Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients: a randomised, double-blind, placebo-controlled trial,” Lancet Lond. Engl., vol. 372, no. 9653, pp. 1906–1913, 29 2008, doi: 10.1016/S0140-6736(08)61525-1.
- S. AB, “Saniona’s tesofensine meets primary and secondary endpoints in Phase 3 obesity registration trial,” GlobeNewswire News Room, Dec. 17, 2018. https://www.globenewswire.com/news-release/2018/12/17/1667781/0/en/Saniona-s-tesofensine-meets-primary-and-secondary-endpoints-in-Phase-3-obesity-registration-trial.html (accessed Nov. 11, 2021).
- A. Sjödin et al., “The effect of the triple monoamine reuptake inhibitor tesofensine on energy metabolism and appetite in overweight and moderately obese men,” Int. J. Obes., vol. 34, no. 11, pp. 1634–1643, Nov. 2010, doi: 10.1038/ijo.2010.87.
- J.-A. Gilbert, C. Gasteyger, A. Raben, D. H. Meier, A. Astrup, and A. Sjödin, “The effect of tesofensine on appetite sensations,” Obes. Silver Spring Md, vol. 20, no. 3, pp. 553–561, Mar. 2012, doi: 10.1038/oby.2011.197.
- A. M. D. Axel, J. D. Mikkelsen, and H. H. Hansen, “Tesofensine, a novel triple monoamine reuptake inhibitor, induces appetite suppression by indirect stimulation of alpha1 adrenoceptor and dopamine D1 receptor pathways in the diet-induced obese rat,” Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol., vol. 35, no. 7, pp. 1464–1476, Jun. 2010, doi: 10.1038/npp.2010.16.
- H. H. Hansen et al., “The novel triple monoamine reuptake inhibitor tesofensine induces sustained weight loss and improves glycemic control in the diet-induced obese rat: comparison to sibutramine and rimonabant,” Eur. J. Pharmacol., vol. 636, no. 1–3, pp. 88–95, Jun. 2010, doi: 10.1016/j.ejphar.2010.03.026.
- Ł. R. Drzyzga, A. Marcinowska, and E. Obuchowicz, “Antiapoptotic and neurotrophic effects of antidepressants: a review of clinical and experimental studies,” Brain Res. Bull., vol. 79, no. 5, pp. 248–257, Jun. 2009, doi: 10.1016/j.brainresbull.2009.03.009.
- S. M. Korte et al., “The many different faces of major depression: it is time for personalized medicine,” Eur. J. Pharmacol., vol. 753, pp. 88–104, Apr. 2015, doi: 10.1016/j.ejphar.2014.11.045.
- D. M. Marks, C.-U. Pae, and A. A. Patkar, “Triple Reuptake Inhibitors: The Next Generation of Antidepressants,” Curr. Neuropharmacol., vol. 6, no. 4, pp. 338–343, Dec. 2008, doi: 10.2174/157015908787386078.
- B. M. Y. Cheung, T. T. Cheung, and N. R. Samaranayake, “Safety of antiobesity drugs,” Ther. Adv. Drug Saf., vol. 4, no. 4, pp. 171–181, Aug. 2013, doi: 10.1177/2042098613489721.
- M. George, M. Rajaram, and E. Shanmugam, “New and emerging drug molecules against obesity,” J. Cardiovasc. Pharmacol. Ther., vol. 19, no. 1, pp. 65–76, Jan. 2014, doi: 10.1177/1074248413501017.
- A. Astrup, S. Madsbad, L. Breum, T. J. Jensen, J. P. Kroustrup, and T. M. Larsen, “Under-reporting of adverse effects of tesofensine,” The Lancet, vol. 382, no. 9887, p. 127, Jul. 2013, doi: 10.1016/S0140-6736(13)61563-9.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.
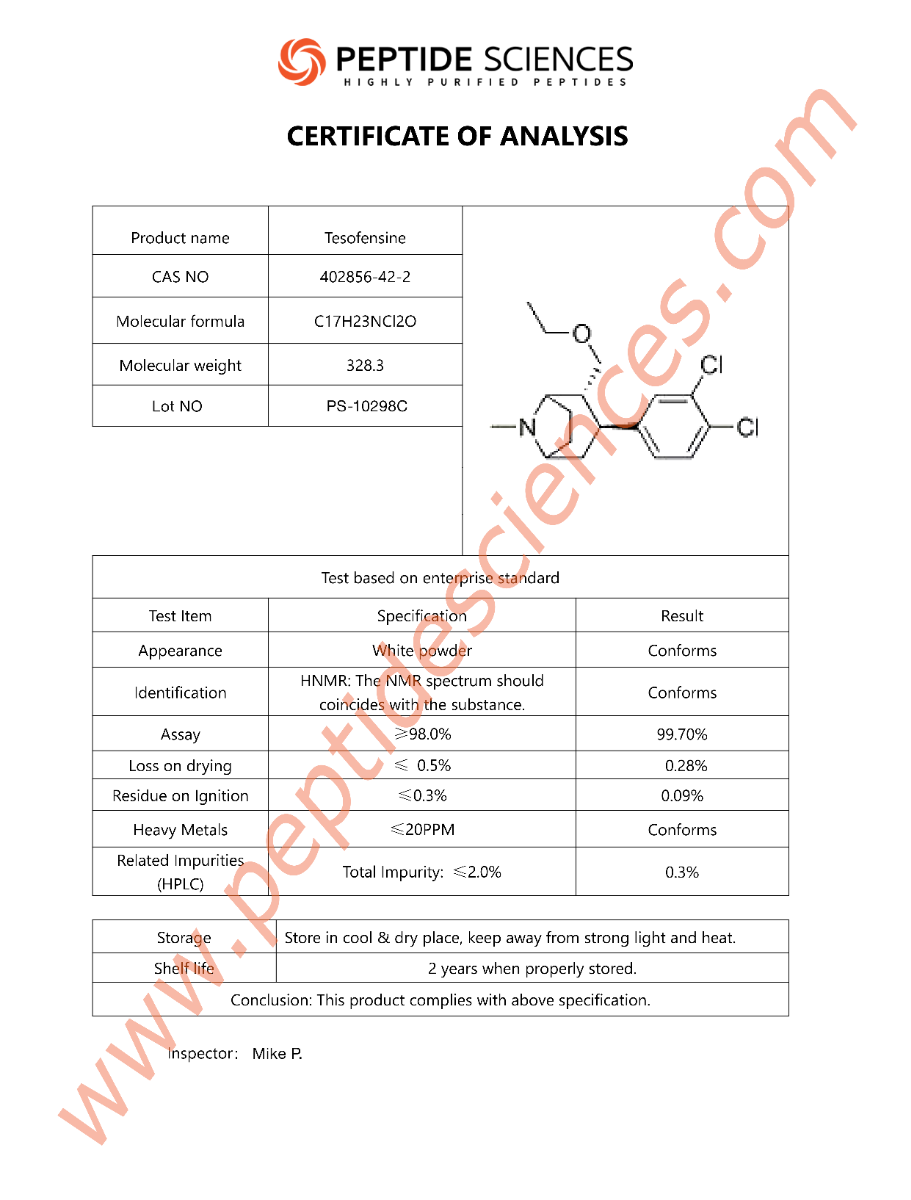
COA
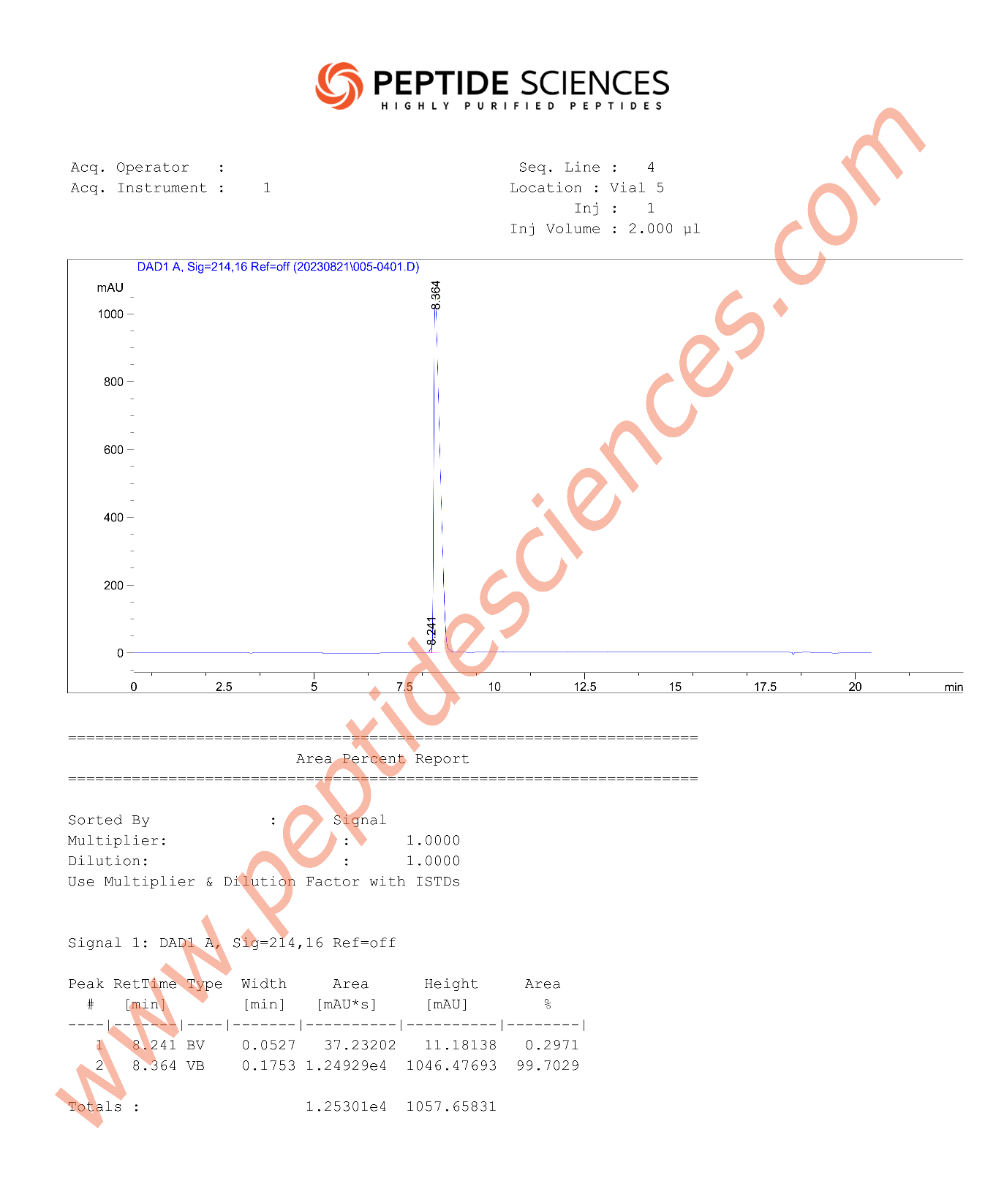
MS
Why Choose Our Tesofensine 500mcg (30 Capsules)?
When you are looking for Tesofensine 500mcg (30 Capsules) for sale, quality is paramount.
Our products undergo rigorous testing to ensure they meet the strict requirements of laboratory environments.
By choosing to buy Tesofensine 500mcg (30 Capsules) from our store, you are guaranteed a product that is:
- High Purity (Tested for 99%+)
- Fast Shipping – Always in stock
- Secure Packaging for Research Integrity
- Competitive Pricing for Bulk Orders
Specifications & Technical Data
| Feature | Specification |
|---|---|
| Product Name | Tesofensine 500mcg (30 Capsules) |
| SKU | 139 |
| Purity | >99% |
| Form | Research Grade Compound |
| Availability | In Stock / For Sale |
Scientific Research & Clinical Applications
The research surrounding Tesofensine 500mcg (30 Capsules) is vast. Scientists explore its potential in various metabolic and physiological models.
For more detailed scientific data, you can visit PubMed
to review the latest peer-reviewed literature regarding this compound.
Frequently Asked Questions
Where can I buy Tesofensine 500mcg (30 Capsules)?
You can buy Tesofensine 500mcg (30 Capsules) directly from our website. We provide a secure checkout and fast shipping to ensure your research stays on track.
Is Tesofensine 500mcg (30 Capsules) in stock?
Yes, we currently have Tesofensine 500mcg (30 Capsules) in stock. Orders are typically processed within 24 hours to ensure rapid delivery to your laboratory.
Related Research Products
If you are interested in Tesofensine 500mcg (30 Capsules), you may also want to explore these related products currently in stock:
Disclaimer: All products listed are for research purposes only. Not for human consumption.
Related products
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides











