Mod GRF, GHRP-2 10mg (Blend)
$95.00
| Properties | |
|---|---|
| Molecular Formula | C121H199N41O40 |
| Molecular Weight | 2868.1 |
| Monoisotopic Mass | 2866.4798003 |
| Polar Area | 1390 |
| Complexity | 6460 |
| XLogP | -25.2 |
| Heavy Atom Count | 202 |
| Hydrogen Bond Donor Count | 49 |
| Hydrogen Bond Acceptor Count | 48 |
| Rotatable Bond Count | 102 |
| PubChem LCSS | MGF Laboratory Chemical Safety Summary |
| Identifiers | |
|---|---|
| CID | 175675731 |
| InChI | InChI=1S/C121H199N41O40/c1-60(166)93(158-108(191)80(55-89(130)173)153-101(184)68(23-8-12-44-123)146-107(190)79(54-88(129)172)155-115(198)95(62(3)168)160-111(194)83(59-165)157-112(195)84-29-18-50-161(84)117(200)85-30-19-51-162(85)116(199)76(36-40-87(128)171)151-96(179)66(126)52-64-31-33-65(169)34-32-64)113(196)150-69(24-9-13-45-124)102(185)156-82(58-164)109(192)149-73(35-39-86(127)170)103(186)144-71(27-16-48-138-120(133)134)99(182)143-70(26-15-47-137-119(131)132)98(181)142-67(22-7-11-43-122)97(180)140-56-90(174)141-81(57-163)110(193)159-94(61(2)167)114(197)154-78(53-63-20-5-4-6-21-63)106(189)148-75(38-42-92(177)178)105(188)147-74(37-41-91(175)176)104(187)145-72(28-17-49-139-121(135)136)100(183)152-77(118(201)202)25-10-14-46-125/h4-6,20-21,31-34,60-62,66-85,93-95,163-169H,7-19,22-30,35-59,122-126H2,1-3H3,(H2,127,170)(H2,128,171)(H2,129,172)(H2,130,173)(H,140,180)(H,141,174)(H,142,181)(H,143,182)(H,144,186)(H,145,187)(H,146,190)(H,147,188)(H,148,189)(H,149,192)(H,150,196)(H,151,179)(H,152,183)(H,153,184)(H,154,197)(H,155,198)(H,156,185)(H,157,195)(H,158,191)(H,159,193)(H,160,194)(H,175,176)(H,177,178)(H,201,202)(H4,131,132,137)(H4,133,134,138)(H4,135,136,139)/t60-,61-,62-,66 ,67 ,68 ,69 ,70 ,71 ,72 ,73 ,74 ,75 ,76 ,77 ,78 ,79 ,80 ,81 ,82 ,83 ,84 ,85 ,93 ,94 ,95 /m1/s1 |
| InChIKey | TXMSCWQMYNJNDC-XGJLPCLZSA-N |
| Isometric SMILES | C[C@H]([C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@@H]2CCCN2C(=O)[C@@H]3CCCN3C(=O)[C@H](CCC(=O)N)NC(=O)[C@H](CC4=CC=C(C=C4)O)N)O |
| Canonical SMILES | CC(C(C(=O)NC(CCCCN)C(=O)NC(CO)C(=O)NC(CCC(=O)N)C(=O)NC(CCCNC(=N)N)C(=O)NC(CCCNC(=N)N)C(=O)NC(CCCCN)C(=O)NCC(=O)NC(CO)C(=O)NC(C(C)O)C(=O)NC(CC1=CC=CC=C1)C(=O)NC(CCC(=O)O)C(=O)NC(CCC(=O)O)C(=O)NC(CCCNC(=N)N)C(=O)NC(CCCCN)C(=O)O)NC(=O)C(CC(=O)N)NC(=O)C(CCCCN)NC(=O)C(CC(=O)N)NC(=O)C(C(C)O)NC(=O)C(CO)NC(=O)C2CCCN2C(=O)C3CCCN3C(=O)C(CCC(=O)N)NC(=O)C(CC4=CC=C(C=C4)O)N)O |
| IUPAC Name | (2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S,3R)-2-[[(2S)-2-[[2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S,3R)-2-[[(2S)-4-amino-2-[[(2S)-6-amino-2-[[(2S)-4-amino-2-[[(2S,3R)-2-[[(2S)-2-[[(2S)-1-[(2S)-1-[(2S)-5-amino-2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]-5-oxopentanoyl]pyrrolidine-2-carbonyl]pyrrolidine-2-carbonyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxybutanoyl]amino]-4-oxobutanoyl]amino]hexanoyl]amino]-4-oxobutanoyl]amino]-3-hydroxybutanoyl]amino]hexanoyl]amino]-3-hydroxypropanoyl]amino]-5-oxopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]hexanoyl]amino]acetyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxybutanoyl]amino]-3-phenylpropanoyl]amino]-4-carboxybutanoyl]amino]-4-carboxybutanoyl]amino]-5-carbamimidamidopentanoyl]amino]hexanoic acid |
Overview
Mechano-growth factor (MGF) is a term used in research literature to describe an IGF-1 splice-isoform context (often referenced as IGF-1Eb/IGF-1Ec family nomenclature depending on species and transcript annotation) and peptide sequences derived from the IGF-1 E-domain region. In laboratory workflows, MGF-related sequences are used as mechanistic probes to interrogate IGF-axis signaling, transcriptional programs associated with cell-cycle progression and differentiation, and injury-response pathway dynamics in controlled in-vitro systems and in-vivo animal models.
A central research theme is that alternative splicing of the IGF-1 locus generates isoform diversity that can be evaluated for differences in expression kinetics, processing, receptor engagement, and downstream signaling outputs under defined experimental conditions.[1]
Biochemical Characteristics
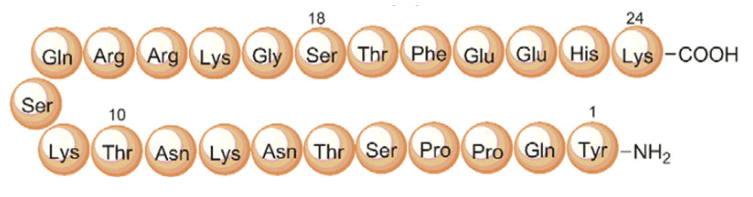
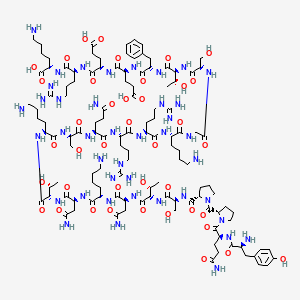
Primary sequence of MGF
Source: ResearchGate
Sequence: Tyr-Gln-Pro-Pro-Ser-Thr-Asn-Lys-Asn-Thr-Lys-Ser-Gln-Arg-Arg-Lys-Gly-Ser-Thr-Phe-Glu-Glu-Arg-Lys-Cys
Molecular Formula: C124H204N42O41S1
Molecular Weight: 2971.99 g/mol
Synonyms: Mechano-growth factor, IGF-1Eb
MGF-related peptides are used in experimental designs as defined-sequence inputs for receptor signaling assays and pathway-mapping studies. Interpretation of outcomes is typically constrained by sequence length, post-translational processing context (isoform-dependent), and the experimental system’s receptor expression profile.
Research Applications
1. IGF-1 Isoform Biology & Alternative Splicing Workflows
MGF is frequently discussed in the context of IGF-1 alternative splicing, where transcript structure and expression kinetics are evaluated across tissues, developmental stages, and perturbation paradigms. Experimental endpoints include isoform-specific mRNA quantification, promoter usage analysis, and mapping of post-translational processing routes that expand IGF-1–related peptide diversity.[1], [2]
2. Muscle Cell Models & Injury-Response Signaling
In skeletal muscle research, MGF-related sequences are used as probes to examine signaling networks associated with myogenic lineage programs in cell culture and animal injury paradigms. Studies often quantify markers of proliferation/differentiation, transcriptional remodeling, and immune-cell–linked resolution signatures in controlled preclinical systems.[3], [4]
3. Mechanotransduction in Cartilage and Intervertebral Disc Models
MGF-related peptides have been used in mechanobiology workflows to map how mechanical overload interfaces with intracellular signaling nodes and migration/apoptosis-associated endpoints in cartilage-relevant and disc-relevant cell systems. These models commonly quantify pathway activation and stress-response signaling under defined mechanical input parameters.[7], [8]
4. Neural and Cardiac Injury Paradigms (Preclinical)
In CNS and cardiac research, MGF/IGF-1 isoform frameworks have been used to explore pathway behavior in ischemia and injury settings in animal models, with endpoints including molecular injury markers, cell survival-associated signaling, and tissue-level functional correlates measured in controlled study designs.[9], [11], [12]
Pathway / Mechanistic Context
Mechanistic studies involving IGF-1 isoforms commonly evaluate signaling through IGF-1 receptor (IGF-1R) and downstream pathways including PI3K/AKT and MAPK/ERK. Depending on model system and experimental design, MGF-associated frameworks may also intersect with mechanotransduction-linked nodes such as RhoA/YAP and stress-activated kinases such as p38 MAPK, particularly in mechanical overload paradigms.[7], [8]
Because IGF-1 isoform nomenclature and processing differ by species and transcript annotation, pathway interpretation in preclinical studies typically includes explicit control of peptide sequence, exposure conditions, receptor expression context, and assay readouts.
Preclinical Research Summary
-
Splicing and isoform regulation: IGF-1 transcript structure and isoform expression profiles vary across developmental stage and experimental perturbations, enabling mechanistic study of transcription/splicing control in aging and tissue maintenance models.[2]
-
Immune-resolution dynamics in muscle injury: Preclinical muscle injury paradigms have evaluated IGF-1 isoform frameworks (including MGF-linked constructs) alongside immune-cell recruitment/resolution markers and cytokine-linked endpoints under controlled conditions.[4]
-
Mechanotransduction in cartilage-relevant systems: Mechanical overload studies in mice have examined MGF-related peptide signaling with mechanistic emphasis on chondrocyte migration behavior and RhoA/YAP-associated pathway outputs.[7]
-
Disc cell stress signaling: Rodent and cell-model work has explored MGF-related peptide exposure in overload-induced apoptosis paradigms, including reported involvement of p38 MAPK signaling endpoints.[8]
-
Neuro and cardiac injury models: Animal studies have reported MGF/IGF-1 isoform-linked observations in ischemia and injury contexts, using molecular and functional readouts to characterize pathway behavior in CNS and myocardium-relevant systems.[9], [11], [12]
Form & Analytical Testing
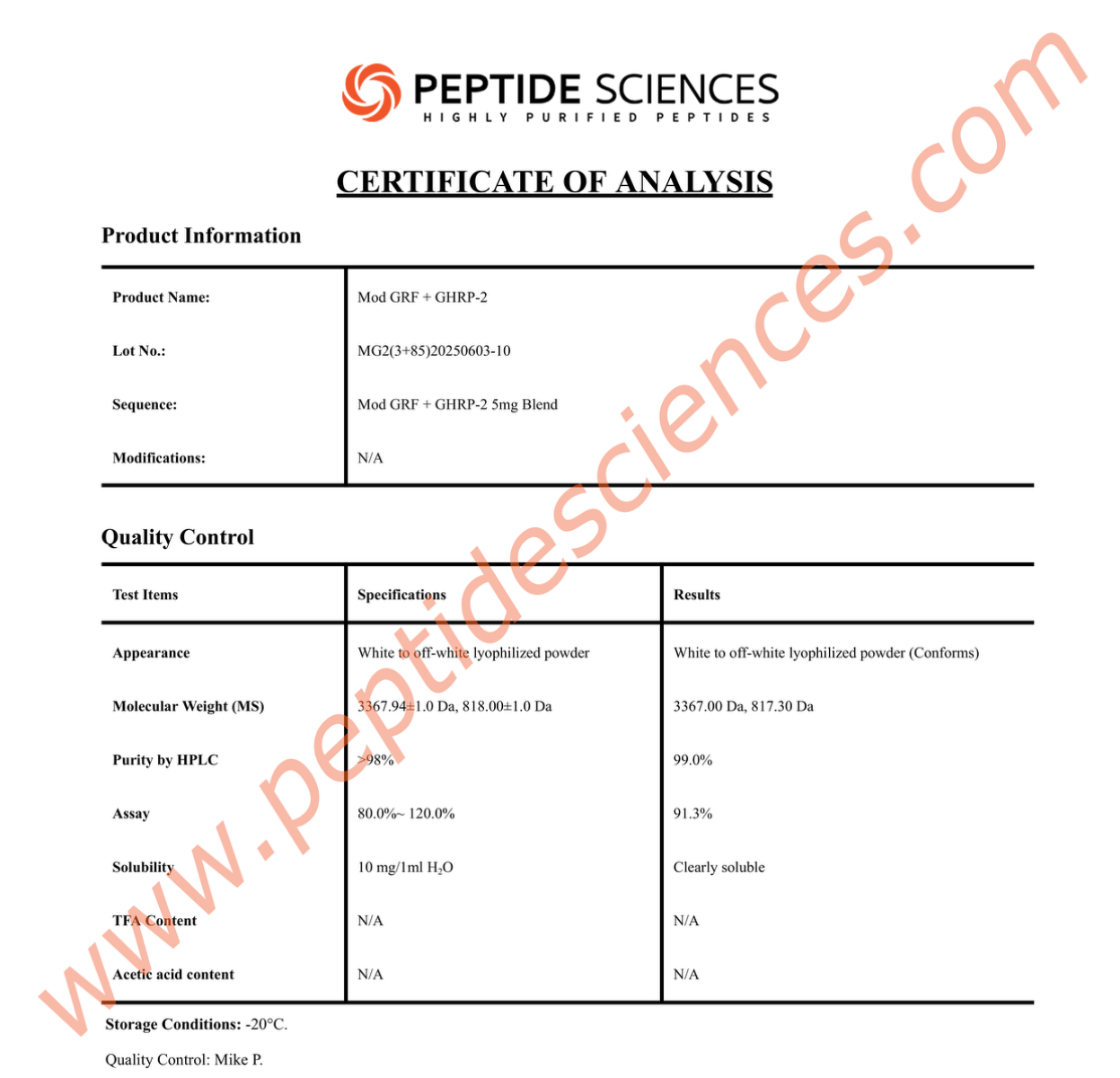
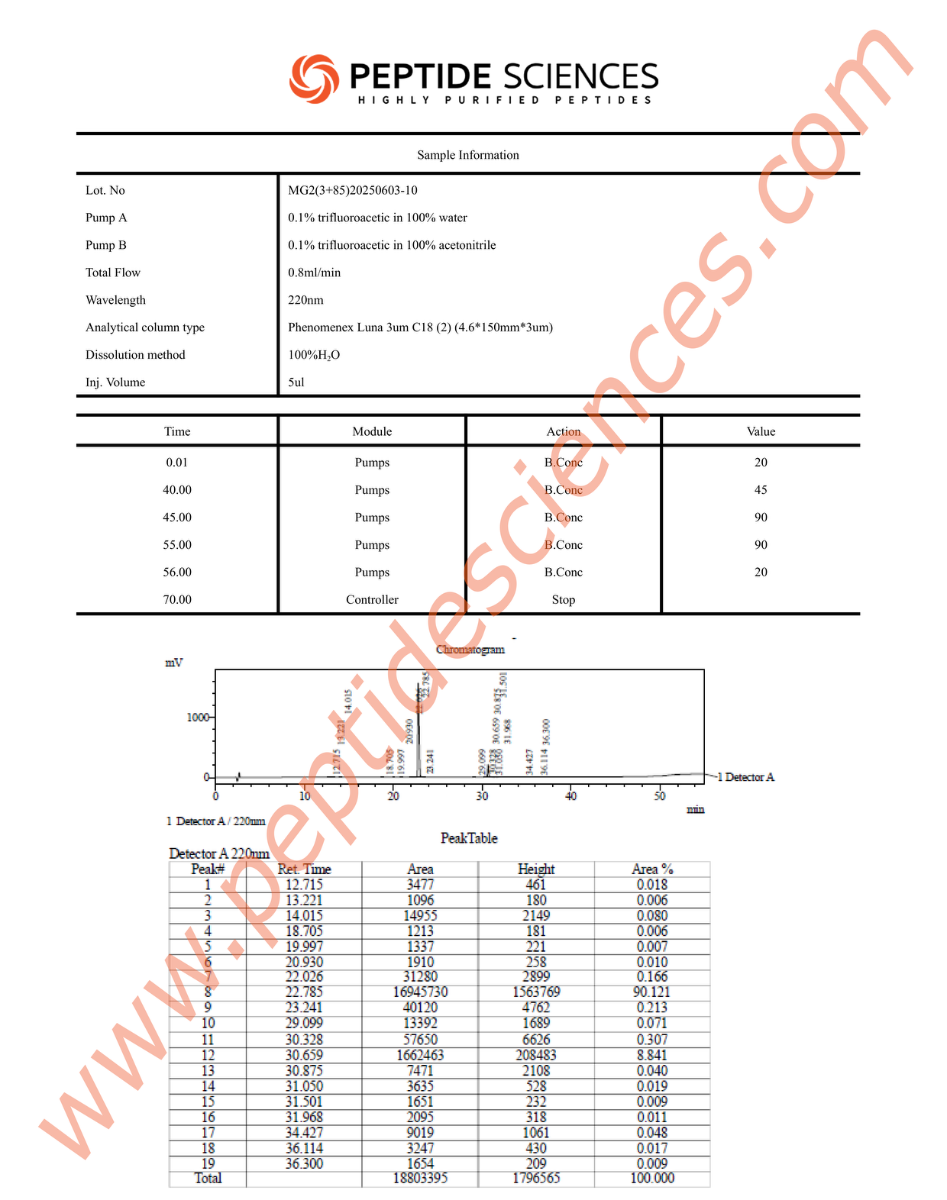
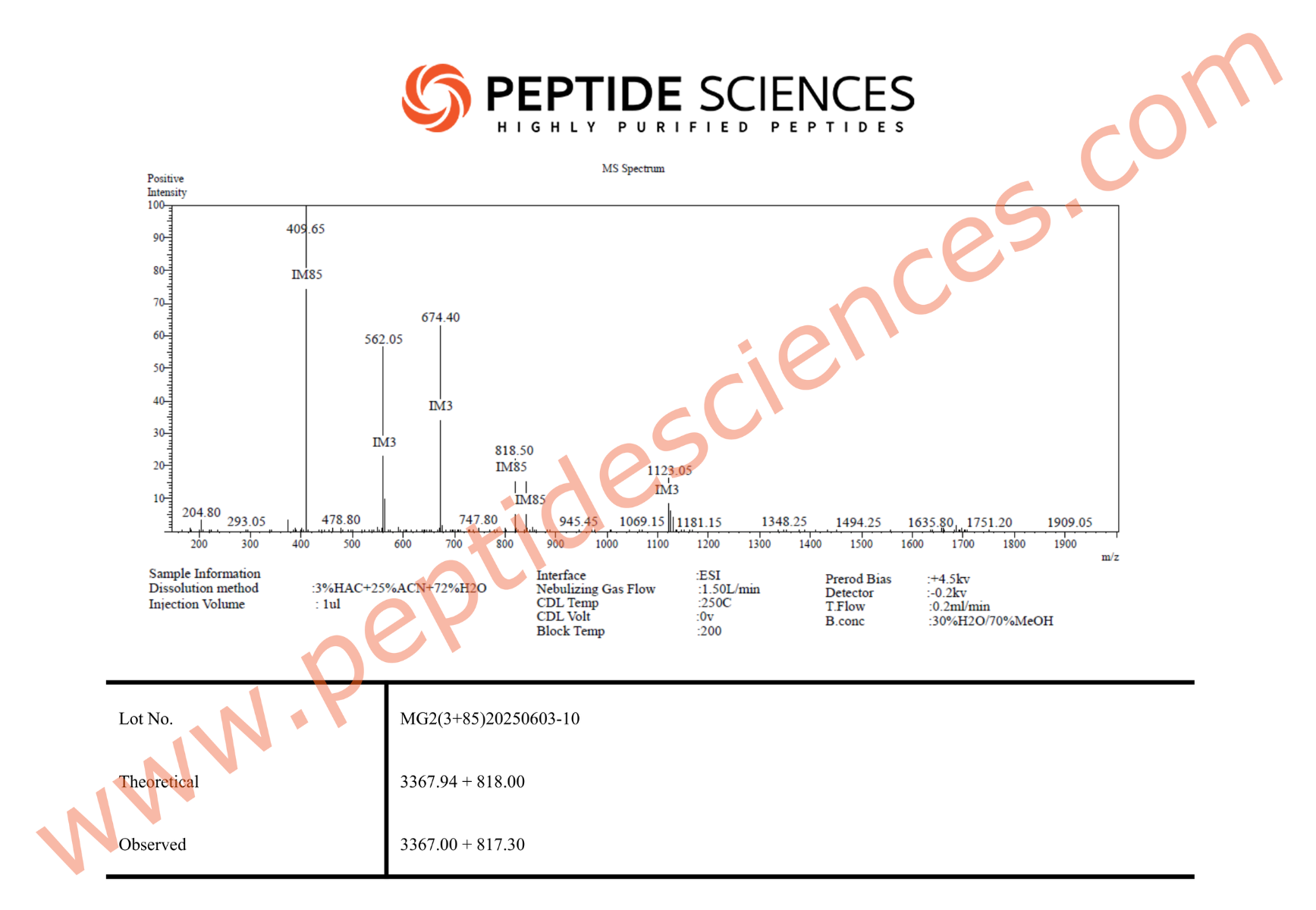
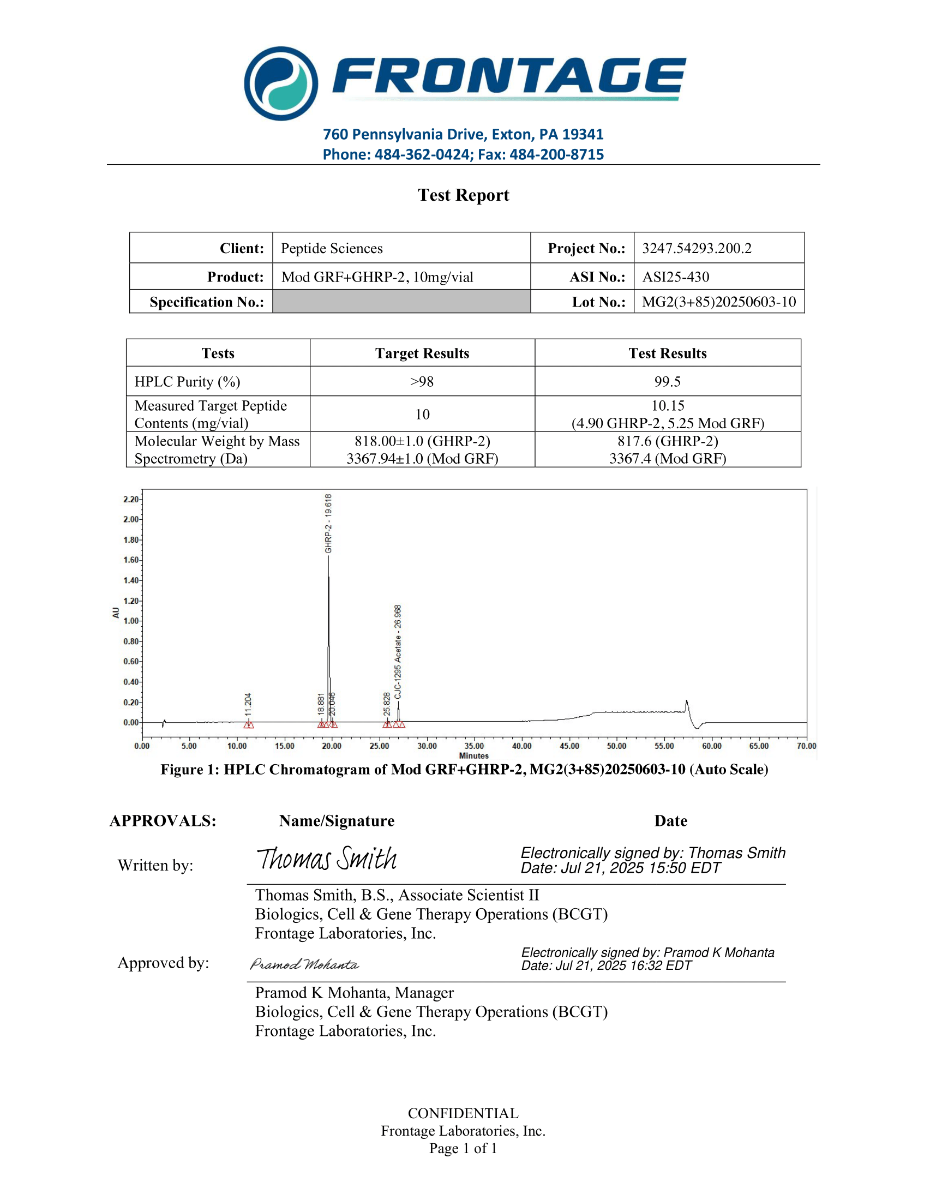
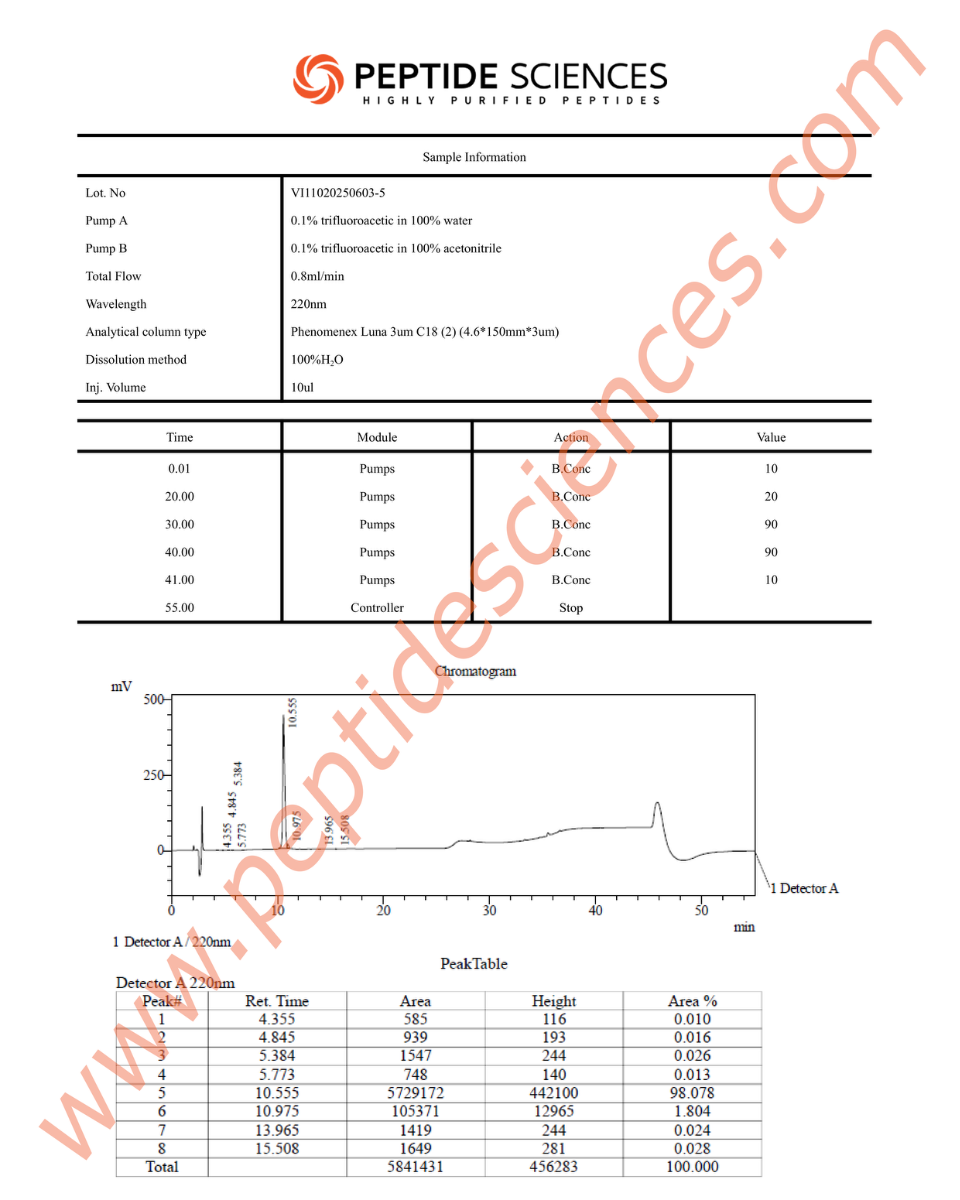
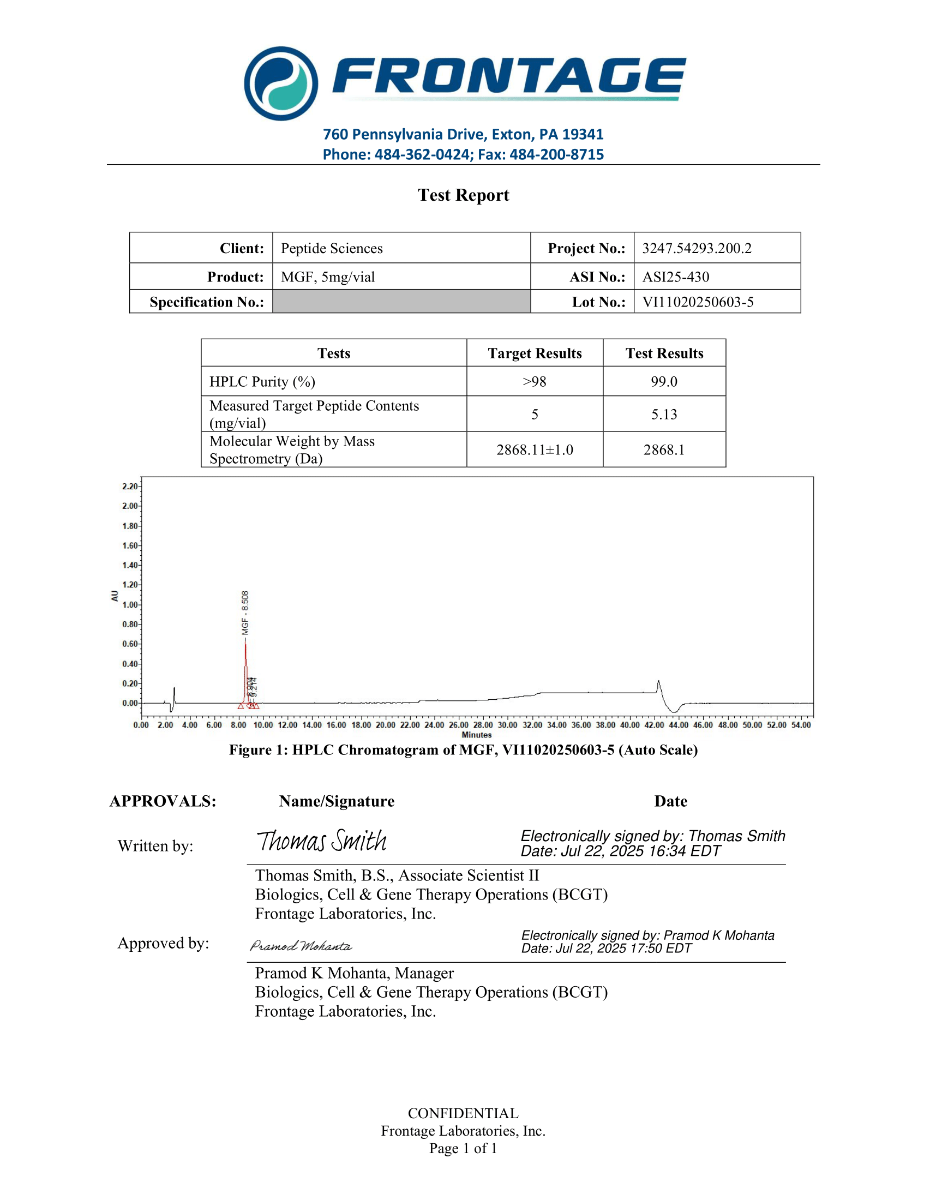
MGF is supplied as a synthetic peptide for laboratory research workflows. Standard quality control for peptide reagents commonly includes chromatographic purity profiling (HPLC) and identity confirmation by mass spectrometry (MS), supported by batch documentation for experimental reproducibility.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Paul Goldspink, PhD is a faculty investigator associated with research examining IGF-1 isoform biology in heart and other tissues, including stress contexts such as mechanical overload, hypoxia, oxidative stress, and aging, with published work that discusses MGF-associated signaling and peptide analogs derived from the E-domain region.
Paul Goldspink, PhD is referenced here to acknowledge published work and research leadership related to IGF-1 isoforms. This reference does not imply endorsement or advocacy of purchase, sale, or use of this product. No affiliation or relationship is implied between the seller and this scientist. Paul Goldspink is cited within the references below.
Referenced Citations
- A. Philippou, M. Maridaki, S. Pneumaticos, and M. Koutsilieris, “The Complexity of the IGF1 Gene Splicing, Posttranslational Modification and Bioactivity,” Mol. Med., vol. 20, no. 1, pp. 202–214, May 2014.
- A. M. Oberbauer, “The Regulation of IGF-1 Gene Transcription and Splicing during Development and Aging,” Front. Endocrinol., vol. 4, Mar. 2013.
- R. W. Matheny, B. C. Nindl, and M. L. Adamo, “Minireview: Mechano-Growth Factor: A Putative Product of IGF-I Gene Expression Involved in Tissue Repair and Regeneration,” Endocrinology, vol. 151, no. 3, pp. 865–875, Mar. 2010.
- K.-T. Sun, K.-K. Cheung, S. W. N. Au, S. S. Yeung, and E. W. Yeung, “Overexpression of Mechano-Growth Factor Modulates Inflammatory Cytokine Expression and Macrophage Resolution in Skeletal Muscle Injury,” Front. Physiol., vol. 9, 2018.
- G. Goldspink, “Research on mechano growth factor: its potential for optimising physical training as well as misuse in doping,” Br. J. Sports Med., vol. 39, no. 11, pp. 787–788, Nov. 2005.
- P. Mills, J. C. Dominique, J. F. Lafrenière, M. Bouchentouf, and J. P. Tremblay, “A Synthetic Mechano Growth Factor E Peptide Enhances Myogenic Precursor Cell Transplantation Success,” Am. J. Transplant., vol. 7, no. 10, pp. 2247–2259, 2007.
- X. Jing et al., “Mechano-growth factor protects against mechanical overload induced damage and promotes migration of growth plate chondrocytes through RhoA/YAP pathway,” Exp. Cell Res., vol. 366, no. 2, pp. 81–91, May 2018.
- Q. Xu, H. Fang, L. Zhao, C. Zhang, L. Zhang, and B. Tian, “Mechano growth factor attenuates mechanical overload-induced nucleus pulposus cell apoptosis through inhibiting the p38 MAPK pathway,” Biosci. Rep., vol. 39, no. 3, Mar. 2019.
- J. Dluzniewska et al., “A strong neuroprotective effect of the autonomous C-terminal peptide of IGF-1 Ec (MGF) in brain ischemia,” FASEB J., vol. 19, no. 13, pp. 1896–1898, Nov. 2005.
- B. Zablocka, P. H. Goldspink, G. Goldspink, and D. C. Gorecki, “Mechano-Growth Factor: an important cog or a loose screw in the repair machinery?,” Front. Endocrinol., vol. 3, 2012.
- J. Riddoch-Contreras, S.-Y. Yang, J. R. T. Dick, G. Goldspink, R. W. Orrell, and L. Greensmith, “Mechano-growth factor, an IGF-I splice variant, rescues motoneurons and improves muscle function in SOD1(G93A) mice,” Exp. Neurol., vol. 215, no. 2, pp. 281–289, Feb. 2009.
- V. Carpenter et al., “Mechano-Growth Factor Reduces Loss of Cardiac Function in Acute Myocardial Infarction,” Heart Lung Circ., vol. 17, no. 1, pp. 33–39, Feb. 2008.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.


Storage Instructions:
All of our products are manufactured using the Lyophilization (Freeze Drying) process, which ensures that our products remain 100% stable for shipping for up to 3-4 months.
Once the peptides are reconstituted (mixed with bacteriostatic water), they must be stored in the fridge to maintain stability. After reconstitution, the peptides will remain stable for up to 30 days.
Lyophilization is a unique dehydration process, also known as cryodesiccation, where the peptides are frozen and then subjected to low pressure. This causes the water in the peptide vial to sublimate directly from solid to gas, leaving behind a stable, crystalline white structure known as lyophilized peptide. The puffy white powder can be stored at room temperature until you’re ready to reconstitute it with bacteriostatic water.
Once peptides have been received, it is imperative that they are kept cold and away from light. If the peptides will be used immediately, or in the next several days, weeks or months, short-term refrigeration under 4C (39F) is generally acceptable. Lyophilized peptides are usually stable at room temperatures for several weeks or more, so if they will be utilized within weeks or months such storage is typically adequate.
However, for longer term storage (several months to years) it is more preferable to store peptides in a freezer at -80C (-112F). When storing peptides for months or even years, freezing is optimal in order to preserve the peptide’s stability.