Follistatin 344 1mg
$175.00
Follistatin 344 is a research peptide variant of the follistatin protein containing 344 amino acids, known to bind myostatin and activin-A, modulating TGF-β signaling. Preclinical studies show it promotes muscle differentiation and regulates growth factor balance via Smad2/3 inhibition. Follistatin 344 is utilized in biochemical and regenerative research investigating peptide–cytokine signaling.
For research use only. Not for human consumption.
References:
Lee SJ et al., Proc Natl Acad Sci USA, 2001 98(16):9306–9311
Cash JN et al., J Biol Chem, 2009 284(13):9011–9020
Sidis Y et al., Endocr Rev, 2001 22(5):726–729
Follistatin 344 1mg – Buy High-Quality Follistatin 344 1mg Online
Looking to buy Follistatin 344 1mg for your research laboratory? You have come to the right place.
We currently have Follistatin 344 1mg for sale and it is in stock and ready for immediate shipping.
Our Follistatin 344 1mg is of the highest purity, making it the best Follistatin 344 1mg online for scientific studies.
Product Overview
Follistatin 344 1mg is a premium research compound widely utilized in various scientific studies.
Researchers seeking to buy Follistatin 344 1mg online often prioritize purity and consistency.
This compound has been studied extensively for its unique biochemical properties and its role in cellular pathways.
Overview
Follistatin (FST) 344 is a synthetic peptide sequence corresponding to a human follistatin isoform, widely used as a laboratory reagent to study ligand sequestration within the transforming growth factor-β (TGF-β) superfamily. In experimental systems, follistatin-derived reagents are used to modulate the extracellular availability of select ligands, including activins and myostatin, enabling mechanistic investigation of downstream signaling behavior under controlled conditions.
Follistatin transcripts and protein isoforms have been detected across multiple tissue contexts, supporting ongoing basic research into isoform-dependent expression patterns and pathway regulation.
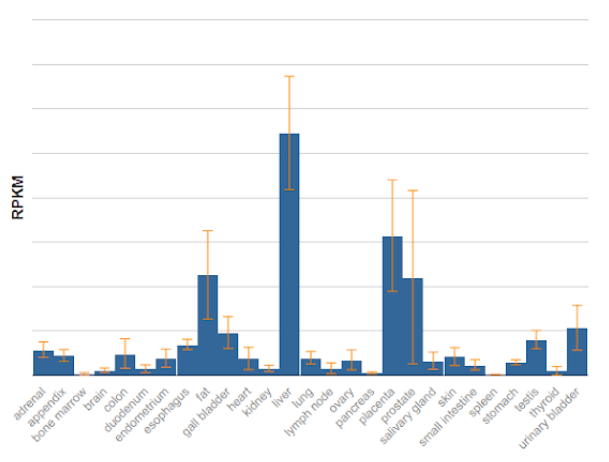
Source: NCBI Gene
Biochemical Characteristics
Source: Uniprot
Sequence: MVRARHQPGG LCLLLLLLCQ FMEDRSAQAG NCWLRQAKNG RCQVLYKTEL SKEECCSTGR LSTSWTEEDV NDNTLFKWMI FNGGAPNCIP CKETCENVDC GPGKKCRMNK KNKPRCVCAP DCSNITWKGP VCGLDGKTYR NECALLKARC KEQPELEVQY QGRCKKTCRD VFCPGSSTCV VDQTNNAYCV TCNRICPEPA SSEQYLCGND GVTYSSACHL RKATCLLGRS IGLAYEGKCI KAKSCEDIQC TGGKKCLWDF KVGRGRCSLC DELCPDSKSD EPVCASDNAT YASECAMKEA ACSSGVLLEV KHSGSCNSIS EDTEEEEEDE DQDYSFPISS ILEW
Molecular Weight: 3780 g/mol
PubChem CID: 178101631
Synonyms: Activin-Binding Protein, FST
Research Applications
- Mechanistic studies of activin neutralization and activin receptor signaling modulation
- Experimental interrogation of myostatin/activin pathway crosstalk within TGF-β superfamily networks
- Cell-based assays evaluating SMAD pathway activation, transcriptional responses, and ligand competition dynamics
- Preclinical model investigations of pathway-regulated tissue remodeling, fibrosis markers, and inflammatory signaling endpoints
- Comparative analyses across follistatin isoforms and engineered variants in controlled laboratory settings
Pathway / Mechanistic Context
Follistatin-associated activity in research settings is generally attributed to high-affinity binding of extracellular ligands (notably activins), limiting ligand availability for receptor engagement and downstream signal propagation. This mechanism is commonly used to study changes in SMAD-dependent transcriptional programs and to map feedback behavior within activin–follistatin regulatory loops.
In addition to activin-related signaling, preclinical literature has investigated follistatin interactions with myostatin and the resulting effects on pathway readouts relevant to ligand sequestration, receptor occupancy, and downstream molecular markers in animal and cellular models[2], [3].
Preclinical Research Summary
Ligand Sequestration Studies in Myostatin/Activin Signaling Models
Animal and cellular studies have used follistatin-based approaches (including protein, mRNA, and gene-expression systems) to evaluate the consequences of myostatin/activin pathway inhibition on molecular markers, tissue morphology, and functional readouts in controlled experimental designs[1], [3], [4], [5].
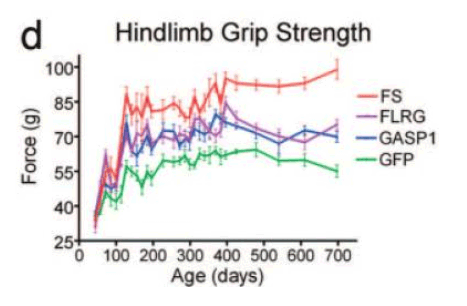
Strength-related readouts reported in follistatin-associated experimental paradigms.
Source: PubMed
Oncology Model Context (Expression and Mechanistic Associations)
Preclinical investigations and molecular profiling studies have examined follistatin expression in tumor-associated contexts and evaluated mechanistic associations with activin-related signaling, migration, and metastatic behavior in defined experimental systems, including mouse models[6], [7], [8], [9].
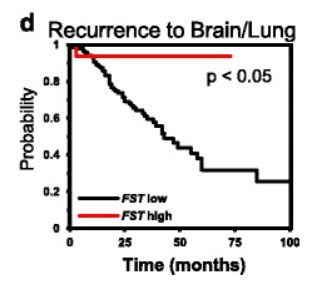
Recurrence-associated probability curves in breast cancer datasets stratified by follistatin (FST) expression.
Source: PubMed
Cell Proliferation, Fibrosis Markers, and Liver Model Systems
Mechanistic research in hepatocyte and hepatic stellate cell biology has examined the activin–follistatin axis in relation to proliferation-associated behavior, apoptosis markers, and fibrosis-related endpoints in controlled animal and cell-based models[10], [11], [12].
Developmental Biology and BMP/TGF-β Signaling Context
Developmental model studies have evaluated TGF-β and BMP signaling dynamics in processes such as optic fissure fusion, providing context for investigating how pathway antagonism and related modulators influence morphogenesis-associated signaling outcomes in model systems[13].
Metabolic and Endocrine Pathway Investigations in Animal Models
Preclinical studies have evaluated forced expression of follistatin in defined animal model systems to assess pathway-regulated molecular and physiological endpoints, including pancreatic β-cell biology and glycemic measurements, under controlled experimental conditions[15].
Note: Citations may include a mixture of preclinical reports and broader pathway reviews. Any cited clinical material is provided solely as bibliographic context and should not be interpreted as suggesting clinical, diagnostic, or therapeutic use.
Form & Analytical Testing
- Supplied as a research-grade reagent intended for controlled laboratory workflows
- Identity and composition supported by analytical characterization (methodology may vary by lot)
- Not manufactured or labeled for diagnostic, therapeutic, clinical, or veterinary applications
Referenced Citations
- C. Schumann et al., “Increasing lean muscle mass in mice via nanoparticle-mediated hepatic delivery of follistatin mRNA,” Theranostics, vol. 8, no. 19, pp. 5276–5288, Oct. 2018.
- L. R. Rodino-Klapac, A. M. Haidet, J. Kota, C. Handy, B. K. Kaspar, and J. R. Mendell, “Inhibition of myostatin with emphasis on follistatin as a therapy for muscle disease,” Muscle Nerve, vol. 39, no. 3, pp. 283–296, Mar. 2009.
- A. Iskenderian et al., “Myostatin and activin blockade by engineered follistatin results in hypertrophy and improves dystrophic pathology in mdx mouse more than myostatin blockade alone,” Skelet. Muscle, vol. 8, Oct. 2018.
- A. M. Haidet et al., “Long-term enhancement of skeletal muscle mass and strength by single gene administration of myostatin inhibitors,” Proc. Natl. Acad. Sci. U. S. A., vol. 105, no. 11, pp. 4318–4322, Mar. 2008.
- C. Barbé et al., “Role of IGF-I in follistatin-induced skeletal muscle hypertrophy,” Am. J. Physiol. – Endocrinol. Metab., vol. 309, no. 6, pp. E557–E567, Sep. 2015.
- C. Zabkiewicz, J. Resaul, R. Hargest, W. G. Jiang, and L. Ye, “Increased Expression of Follistatin in Breast Cancer Reduces Invasiveness and Clinically Correlates with Better Survival,” Cancer Genomics Proteomics, vol. 14, no. 4, pp. 241–251, Aug. 2017.
- D. D. Seachrist, S. T. Sizemore, E. Johnson, F. W. Abdul-Karim, K. L. Weber Bonk, and R. A. Keri, “Follistatin is a metastasis suppressor in a mouse model of HER2-positive breast cancer,” Breast Cancer Res. BCR, vol. 19, no. 1, p. 66, 05 2017.
- E. Bloise et al., “Differential expression of follistatin and FLRG in human breast proliferative disorders,” BMC Cancer, vol. 9, p. 320, Sep. 2009.
- L. SHI, J. RESAUL, S. OWEN, L. YE, and W. G. JIANG, “Clinical and Therapeutic Implications of Follistatin in Solid Tumours,” Cancer Genomics Proteomics, vol. 13, no. 6, pp. 425–436, Oct. 2016.
- H. Ooe et al., “Proliferation of rat small hepatocytes requires follistatin expression,” J. Cell. Physiol., vol. 227, no. 6, pp. 2363–2370, Jun. 2012.
- S. Patella, D. J. Phillips, J. Tchongue, D. M. de Kretser, and W. Sievert, “Follistatin attenuates early liver fibrosis: effects on hepatic stellate cell activation and hepatocyte apoptosis,” Am. J. Physiol. Gastrointest. Liver Physiol., vol. 290, no. 1, pp. G137-144, Jan. 2006.
- M. Grusch et al., “Deregulation of the activin/follistatin system in hepatocarcinogenesis,” J. Hepatol., vol. 45, no. 5, pp. 673–680, Nov. 2006.
- M. D. Knickmeyer et al., “TGFβ-facilitated optic fissure fusion and the role of bone morphogenetic protein antagonism,” Open Biol., vol. 8, no. 3, 2018.
- M. P. Zimber et al., “Hair regrowth following a Wnt- and follistatin containing treatment: safety and efficacy in a first-in-man phase 1 clinical trial,” J. Drugs Dermatol. JDD, vol. 10, no. 11, pp. 1308–1312, Nov. 2011.
- C. Zhao et al., “Overcoming Insulin Insufficiency by Forced Follistatin Expression in β-cells of db/db Mice,” Mol. Ther., vol. 23, no. 5, pp. 866–874, May 2015.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.
Storage Instructions:
All of our products are manufactured using the Lyophilization (Freeze Drying) process, which ensures that our products remain 100% stable for shipping for up to 3-4 months.
Once the peptides are reconstituted (mixed with bacteriostatic water), they must be stored in the fridge to maintain stability. After reconstitution, the peptides will remain stable for up to 30 days.
Lyophilization is a unique dehydration process, also known as cryodesiccation, where the peptides are frozen and then subjected to low pressure. This causes the water in the peptide vial to sublimate directly from solid to gas, leaving behind a stable, crystalline white structure known as lyophilized peptide. The puffy white powder can be stored at room temperature until you’re ready to reconstitute it with bacteriostatic water.
Once peptides have been received, it is imperative that they are kept cold and away from light. If the peptides will be used immediately, or in the next several days, weeks or months, short-term refrigeration under 4C (39F) is generally acceptable. Lyophilized peptides are usually stable at room temperatures for several weeks or more, so if they will be utilized within weeks or months such storage is typically adequate.
However, for longer term storage (several months to years) it is more preferable to store peptides in a freezer at -80C (-112F). When storing peptides for months or even years, freezing is optimal in order to preserve the peptide’s stability.
For further information on proper storage techniques, click the link below:
Peptide Storage Information
What is Follistatin?
Follistatin was first identified in the late 1980s, when researchers discovered that it could block the activity of myostatin, a protein that regulates muscle growth. This finding sparked a great deal of interest in the scientific community, and over the next several years, researchers began to uncover more information about the protein’s structure and function.
One of the key breakthroughs came in 1997, when scientists discovered that a genetic mutation in a breed of cattle called the “Belgian Blue” led to a dramatic increase in muscle mass due to a mutation in the myostatin gene. This study was significant because it helped to confirm the role of myostatin as a “brake” on muscle growth, and it also highlighted the potential of inhibiting its activity to increase muscle mass. Research into follistatin continued over the next several years, leading to the development of recombinant forms of the protein that could be used to supplement the levels in the body.
In short, follistatin is a protein that plays a critical role in regulating muscle growth and development. It acts as a natural antagonist to myostatin, a protein that limits muscle growth, by binding to and inhibiting its function. This makes follistatin a potentially powerful tool for athletes, bodybuilders, and anyone looking to increase muscle mass and strength. In this post, we’ll explore the science behind follistatin and its potential benefits.
Why Choose Our Follistatin 344 1mg?
When you are looking for Follistatin 344 1mg for sale, quality is paramount.
Our products undergo rigorous testing to ensure they meet the strict requirements of laboratory environments.
By choosing to buy Follistatin 344 1mg from our store, you are guaranteed a product that is:
- High Purity (Tested for 99%+)
- Fast Shipping – Always in stock
- Secure Packaging for Research Integrity
- Competitive Pricing for Bulk Orders
Specifications & Technical Data
| Feature | Specification |
|---|---|
| Product Name | Follistatin 344 1mg |
| SKU | 38 |
| Purity | >99% |
| Form | Research Grade Compound |
| Availability | In Stock / For Sale |
Scientific Research & Clinical Applications
The research surrounding Follistatin 344 1mg is vast. Scientists explore its potential in various metabolic and physiological models.
For more detailed scientific data, you can visit PubMed
to review the latest peer-reviewed literature regarding this compound.
Frequently Asked Questions
Where can I buy Follistatin 344 1mg?
You can buy Follistatin 344 1mg directly from our website. We provide a secure checkout and fast shipping to ensure your research stays on track.
Is Follistatin 344 1mg in stock?
Yes, we currently have Follistatin 344 1mg in stock. Orders are typically processed within 24 hours to ensure rapid delivery to your laboratory.
Related Research Products
If you are interested in Follistatin 344 1mg, you may also want to explore these related products currently in stock:
Disclaimer: All products listed are for research purposes only. Not for human consumption.
Related products
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides