Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment)
$300.00
Repair and Recovery is a research peptide formulation containing stable BPC-157 Arginate and Thymosin Beta-4 fragment designed to study molecular regeneration and tissue remodeling mechanisms. Preclinical data indicate synergistic modulation of angiogenic, cytoprotective, and actin-polymerization pathways through nitric oxide and growth factor signaling. This combination provides a model for investigating cellular repair, wound modulation, and peptide-mediated recovery dynamics in controlled laboratory conditions.
References:
Sikiric P et al., Curr Pharm Des, 2018 24(18):2002–2030
Goldstein AL et al., Ann NY Acad Sci, 2007 1112:148–154
Malinda KM et al., FASEB J, 1999 13(14):2106–2114
These capsules are provided exclusively as inert containers for the dry transport, storage, and handling of laboratory research materials. They are not dosage forms and are not intended for ingestion or administration. They must not be used as drug delivery systems, consumed, or administered in any form. For Research Use Only: Not for use in humans or animals. Not for diagnostic or therapeutic purposes.
Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) – Buy High-Quality Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) Online
Looking to buy Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) for your research laboratory? You have come to the right place.
We currently have Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) for sale and it is in stock and ready for immediate shipping.
Our Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) is of the highest purity, making it the best Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) online for scientific studies.
Product Overview
Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) is a premium research compound widely utilized in various scientific studies.
Researchers seeking to buy Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) online often prioritize purity and consistency.
This compound has been studied extensively for its unique biochemical properties and its role in cellular pathways.
Overview
This page presents a laboratory-focused overview of two research-use peptide ingredients: BPC-157 (Arginate Salt) and a Thymosin Beta-4–derived fragment, Ac-SDKP (Ac-Ser-Asp-Lys-Pro). In preclinical research settings, these peptides are evaluated as biochemical tools for studying cytoskeletal dynamics, cell migration signaling, extracellular matrix (ECM) organization, and vascular-associated pathway activity in controlled in-vitro, ex-vivo, and in-vivo animal models.
The content below is written for mechanistic interpretation of published non-clinical studies and is limited to molecular, cellular, and animal research contexts. No statements are made regarding safety, efficacy, or intended use beyond research workflows.
Biochemical Characteristics
BPC-157 (Arginate Salt): BPC-157 is a synthetic pentadecapeptide derived from a larger gastric peptide sequence referred to in the literature as Body Protection Compound (BPC). In preclinical models, BPC-157 is studied using endpoints that include fibroblast-associated signaling markers, ECM-related gene/protein expression panels, nitric oxide–linked pathway activity, and growth factor receptor expression profiles. Salt form selection (e.g., arginate vs acetate) is evaluated experimentally through physicochemical stability testing, degradation kinetics in simulated matrices, and analytical recovery under defined storage and assay conditions.
Thymosin Beta-4 Fragment (Ac-SDKP): Ac-SDKP (Ac-Ser-Asp-Lys-Pro) is a tetrapeptide generated endogenously via proteolytic processing of Thymosin Beta-4. In experimental systems, Ac-SDKP is used to interrogate actin-linked processes (via upstream Thymosin Beta-4 biology), endothelial signaling and migration indices, and fibroblast lineage-state markers under controlled stimulation paradigms. Ac-SDKP is also studied as a substrate of angiotensin-converting enzyme (ACE) in biochemical and structural enzymology contexts.
Context on Thymosin Beta-4 (parent peptide, not an ingredient): Thymosin Beta-4 is a 43–amino acid peptide recognized as a principal actin-sequestering protein in many cell types. Mechanistically, Thymosin Beta-4 binds monomeric actin and influences actin filament polymerization, cytoskeletal turnover, and cell motility. Fragment-based research (including Ac-SDKP) is commonly used to isolate discrete molecular functions and to evaluate size-dependent transport and stability properties in experimental models.
Research Applications
- Actin dynamics and cytoskeletal turnover assays (e.g., actin polymerization state markers; motility indices)
- Cell migration pathway studies in endothelial and fibroblast models (e.g., wound-scratch assays, transwell migration; signaling phosphorylation panels)
- Extracellular matrix organization and remodeling readouts (e.g., collagen-related transcriptional panels; matrix deposition quantification; histology in animal models)
- Nitric oxide–associated signaling interrogation (e.g., NOS-linked markers, downstream pathway activation states)
- ACE–substrate enzymology and peptide hydrolysis studies for Ac-SDKP (biochemical kinetics; structural binding context)
- Salt form and stability comparisons (arginate vs acetate) using forced-degradation and simulated matrix exposure protocols
Pathway / Mechanistic Context
Thymosin Beta-4–derived peptide fragments, including Ac-SDKP, are utilized in experimental systems to examine actin-associated processes such as actin monomer sequestration and cytoskeletal turnover. These activities are evaluated using molecular and cellular assays without reference to organism-level function.
Ac-SDKP is further investigated in preclinical models for its role in endothelial cell signaling and fibroblast lineage regulation, with endpoints limited to transcriptional markers, migration indices, and pathway activation states. Ac-SDKP is also characterized as an ACE substrate, and ACE-dependent hydrolysis is used as a mechanistic axis in structural and enzymology studies.
BPC-157 is reported in preclinical literature to interact with nitric oxide–associated signaling pathways, growth factor receptor expression, and extracellular matrix–related molecular processes. These interactions are characterized using biochemical assays, histological analysis, and pathway-level measurements in cellular and animal models.
When evaluated within the same experimental framework, these peptides serve as independent molecular probes for investigating distinct biochemical pathways related to cytoskeletal dynamics and extracellular matrix regulation.
Preclinical Research Summary
Published animal and cellular studies involving BPC-157 commonly report pathway-level changes quantified by (i) connective tissue cell migration indices, (ii) fibroblast-associated transcriptional programs, (iii) extracellular matrix composition markers, and (iv) vascular-associated signaling panels. Experimental designs typically rely on histological scoring, immunohistochemistry, gene/protein expression profiling, and biochemical marker quantification.
Preclinical investigations involving Ac-SDKP frequently evaluate endothelial signaling and migration readouts, fibroblast differentiation-state markers, and stress-response pathway activation states in defined stimulation models. In parallel, ACE-mediated hydrolysis of Ac-SDKP is studied using biochemical kinetics and structural characterization approaches, providing a mechanistic basis for interpreting Ac-SDKP availability in experimental systems.
The mechanistic literature often positions these peptides in adjacent experimental themes (cell motility, matrix organization, vascular-associated signaling). Combined evaluation, where performed, is interpreted through discrete molecular endpoints rather than organism-level functional claims.
Ac-SDKP (TB-500 Fragment)
Sequence: Ac-SDKP
Molecular Formula: C20H33N5O9
Molecular Weight: 487.5 g/mol
PubChem CID: 65938
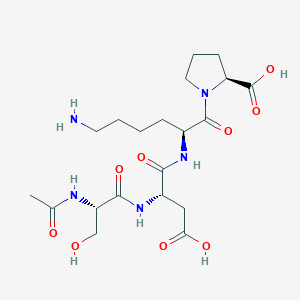
Source: PubChem
Salt-form discussions in the BPC-157 literature are typically framed around stability and degradation kinetics measured under controlled laboratory conditions (e.g., temperature stress, light exposure, and simulated matrix exposure). These experiments are used to compare analytical recovery and integrity of different salt forms under defined handling and assay workflows.
Form & Analytical Testing
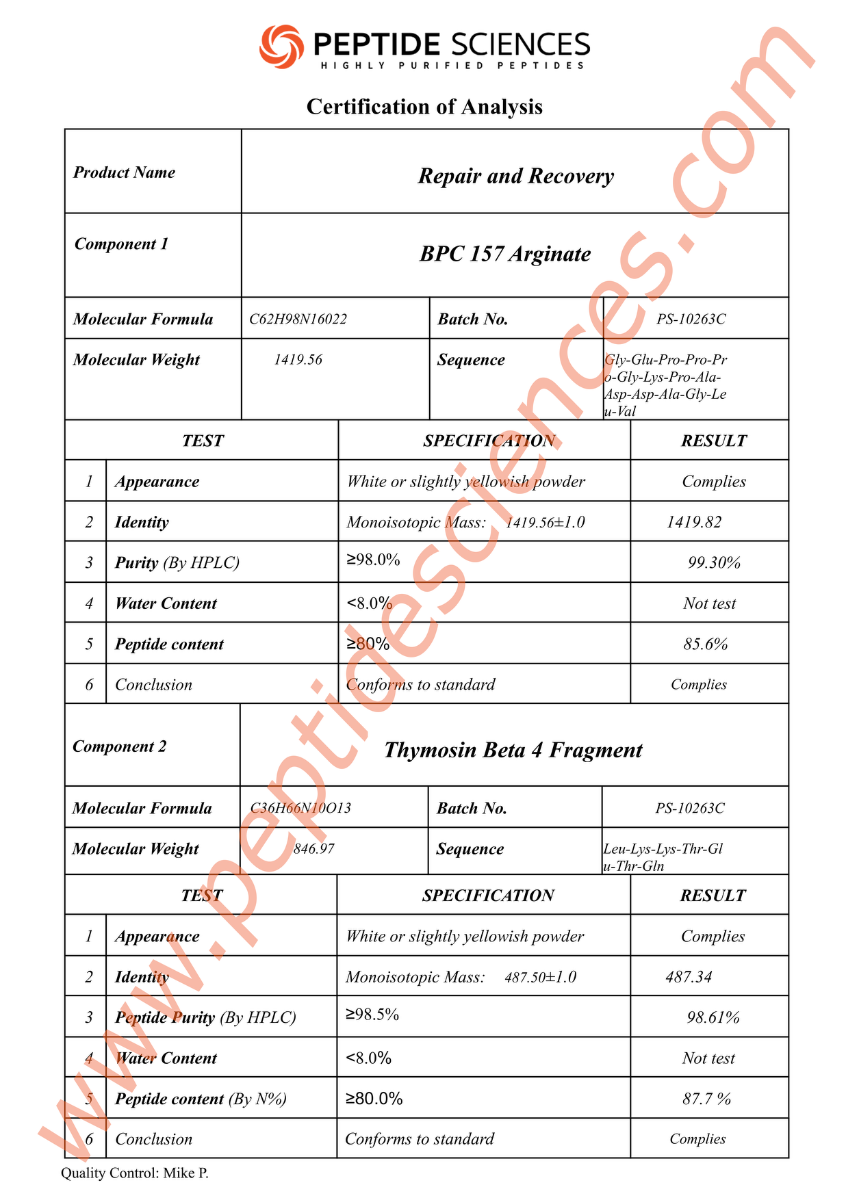
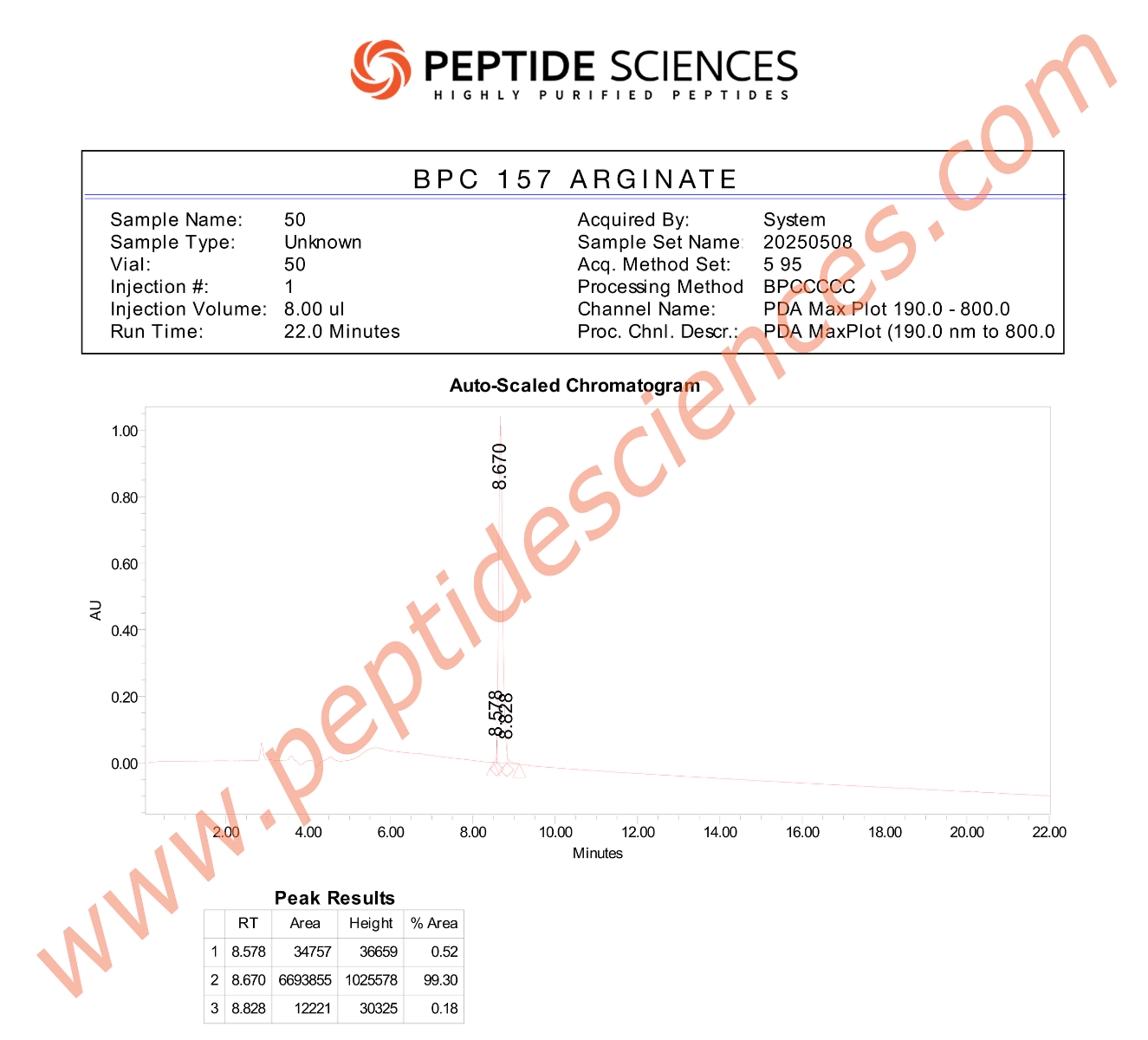
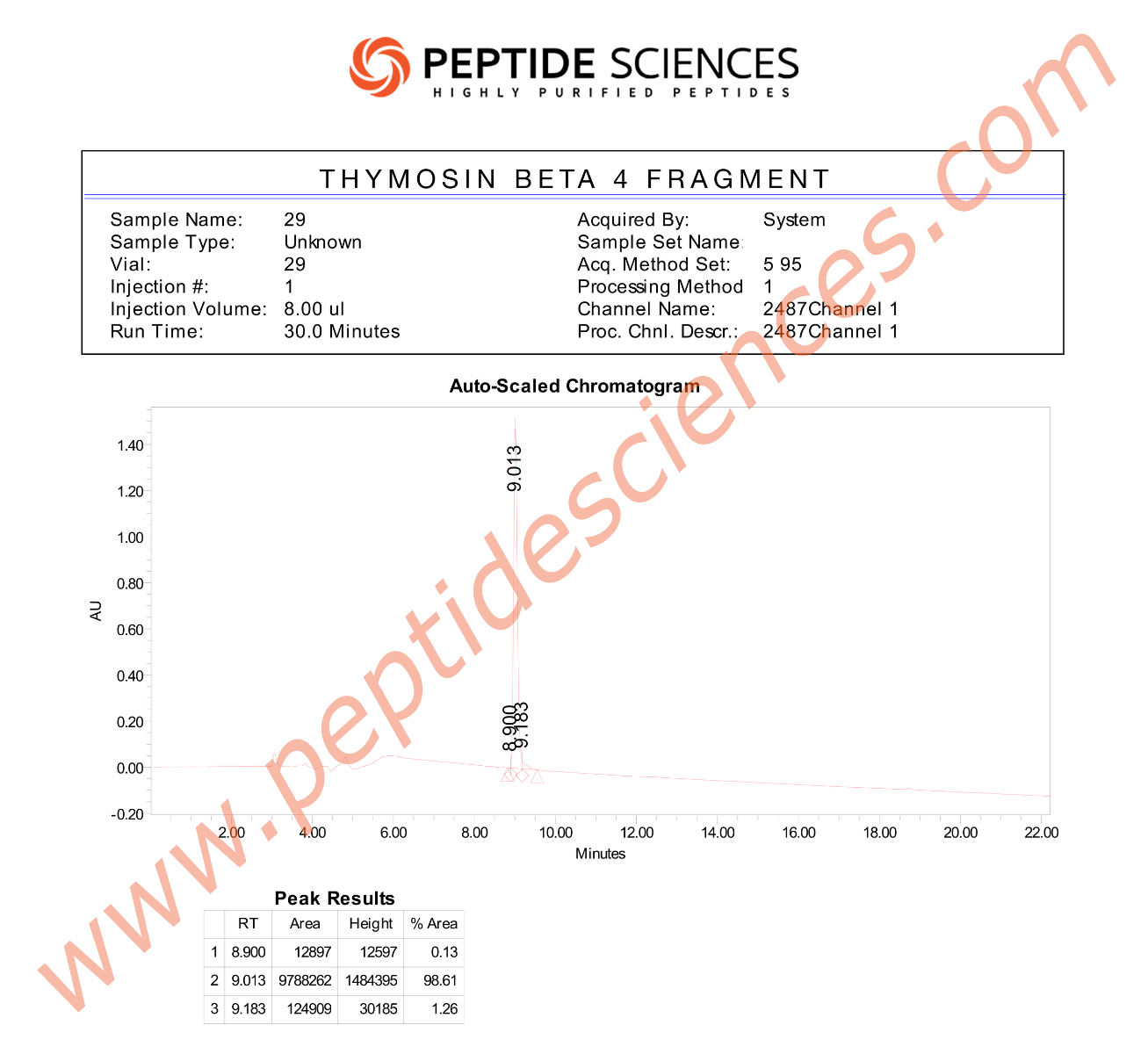
Research-grade peptide materials are typically characterized using analytical methods such as HPLC/UPLC for purity determination, LC-MS for molecular mass confirmation, and (where applicable) NMR for structural verification. Lot-specific Certificates of Analysis (COA) commonly report identity confirmation, chromatographic purity, and additional quality attributes according to internal laboratory standards and applicable research protocols.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Allan L. Goldstein, MD, Allan L. Goldstein is professor and Catharine B. & William McCormick Chair of the department of Biochemistry and Molecular Biology at The George Washington University School of Medicine and Health Sciences, where he has served since 1978. Thymosins were discovered in the mid 1960’s, when Allan Goldstein from the Laboratory of Abraham White at the Albert Einstein College of Medicine in New York studied the role of the thymus in development of the vertebrate immune system. He is a world-renowned authority on the thymus gland and the workings of the immune system, and co-discoverer of the thymosins. Dr. Goldstein is the author of over 400 scientific articles in professional journals, the inventor on more than 15 U.S. Patents, and the editor of several books in the fields of biochemistry, biomedicine, immunology and neuro-science. He is on the editorial boards of numerous scientific and medical journals and has been a consultant to many re-search organizations in industry and government; co-founder of The Institute for Advanced Studies in Aging and Geriatric Medicine, a non-profit research and educational institute; a member of the Board of Trustees of the Albert Sabin Vaccine Institute; and serves as the Chairman of the Board of RegeneRx Biopharmaceuticals. Dr. Goldstein received his B.S. from Wagner College in 1959 and his M.S. and Ph.D. from Rutgers University in 1964. He served as a faculty member of the Albert Einstein College of Medicine from 1964 to 1972, and moved to the University of Texas Medical Branch in Galveston in 1972 as professor and director of the division of Biochemistry.
Allan L. Goldstein, MD is being referenced as one of the leading scientists involved in the research and development of TB4, TB-500 and other Thymosins. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Goldstein is listed in [10] under the referenced citations.
Referenced Citations
- R. Rucman, “Stable pentadecapeptide salts, a process for preparation thereof, a use thereof in the manufacture of pharmaceutical preparations and a use thereof in therapy,” US9850282B2, Dec. 26, 2017.
- A. Duzel et al., “Stable gastric pentadecapeptide BPC 157 in the treatment of colitis and ischemia and reperfusion in rats: New insights,” World J. Gastroenterol., vol. 23, no. 48, pp. 8465–8488, Dec. 2017, doi: 10.3748/wjg.v23.i48.8465.
- M. Chopp and Z. G. Zhang, “Thymosin β4 as a restorative/regenerative therapy for neurological injury and neurodegenerative diseases,” Expert Opin. Biol. Ther., vol. 15, no. sup1, Art. no. sup1, Jun. 2015, doi: 10.1517/14712598.2015.1005596.
- “Thymosin Beta-4 Molecule Prompts Damaged Cells To Repair Themselves After Heart Attack,” Science 2.0, Aug. 27, 2014. http://www.science20.com/news_releases/thymosin_beta4_molecule_prompts_damaged_cells_repair_themselves_after_heart_attack (accessed Dec. 02, 2015).
- C.-H. Chang, W.-C. Tsai, M.-S. Lin, Y.-H. Hsu, and J.-H. S. Pang, “The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration,” J. Appl. Physiol. Bethesda Md 1985, vol. 110, no. 3, pp. 774–780, Mar. 2011, doi: 10.1152/japplphysiol.00945.2010.
- S. Pejman, M. Kamarehei, G. Riazi, S. Pooyan, and S. Balalaie, “Ac-SDKP ameliorates the progression of experimental autoimmune encephalomyelitis via inhibition of ER stress and oxidative stress in the hippocampus of C57BL/6 mice,” Brain Res. Bull., vol. 154, pp. 21–31, Jan. 2020, doi: 10.1016/j.brainresbull.2019.09.014.
- D. Gwyer, N. M. Wragg, and S. L. Wilson, “Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing,” Cell Tissue Res., vol. 377, no. 2, pp. 153–159, Aug. 2019, doi: 10.1007/s00441-019-03016-8.
- T. Cerovecki et al., “Pentadecapeptide BPC 157 (PL 14736) improves ligament healing in the rat,” J. Orthop. Res. Off. Publ. Orthop. Res. Soc., vol. 28, no. 9, pp. 1155–1161, Sep. 2010, doi: 10.1002/jor.21107.
- C.-H. Chang, W.-C. Tsai, Y.-H. Hsu, and J.-H. S. Pang, “Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts,” Mol. Basel Switz., vol. 19, no. 11, pp. 19066–19077, Nov. 2014, doi: 10.3390/molecules191119066.
- Song, Ran & Choi, Hyun & Yang, Hyung-In & Yoo, Myung & Park, Yong-Beom & Kim, Kyoung. (2012). Association between serum thymosin β4 levels of rheumatoid arthritis patients and disease activity and response to therapy. Clinical rheumatology. 31. 1253-8. 10.1007/s10067-012-2011-7. [Research Gate]
Ac-SDKP (Thymosin Beta4 Fragment) Oral Bioavailability Research Studies
- Masuyer, Geoffrey, et al. “Structural Basis of Ac-SDKP Hydrolysis by Angiotensin-I Converting Enzyme.” Scientific Reports, vol. 5, no. 1, 25 Sept. 2015, 10.1038/srep13742.
- Nakagawa, Pablo, et al. “Ac-SDKP Decreases Mortality and Cardiac Rupture after Acute Myocardial Infarction.” PloS One, vol. 13, no. 1, 2018, p. e0190300, pubmed.ncbi.nlm.nih.gov/29364896/, 10.1371/journal.pone.0190300.
- Jin, Fuyu, et al. “Ac-SDKP Attenuates Activation of Lung Macrophages and Bone Osteoclasts in Rats Exposed to Silica by Inhibition of TLR4 and RANKL Signaling Pathways.” Journal of Inflammation Research, vol. 14, 2021, pp. 1647–1660, pubmed.ncbi.nlm.nih.gov/33948088/, 10.2147/JIR.S306883.
- Xu, Hong, et al. “A New Antifibrotic Target of Ac-SDKP: Inhibition of Myofibroblast Differentiation in Rat Lung with Silicosis.” PLoS ONE, vol. 7, no. 7, 3 July 2012, p. e40301, 10.1371/journal.pone.0040301.
- Srivastava, Swayam Prakash, et al. “Inhibition of Angiotensin-Converting Enzyme Ameliorates Renal Fibrosis by Mitigating DPP-4 Level and Restoring Antifibrotic MicroRNAs.” Genes, vol. 11, no. 2, 18 Feb. 2020, p. E211, pubmed.ncbi.nlm.nih.gov/32085655/, 10.3390/genes11020211.
- Shifeng, Li, et al. “Ac-SDKP Increases α-TAT 1 and Promotes the Apoptosis in Lung Fibroblasts and Epithelial Cells Double-Stimulated with TGF-β1 and Silica.” Toxicology and Applied Pharmacology, vol. 369, 15 Apr. 2019, pp. 17–29, www.sciencedirect.com/science/article/abs/pii/S0041008X19300729, 10.1016/j.taap.2019.02.015.
- Kumar, Nitin, and Congcong Yin. “The Anti-Inflammatory Peptide Ac-SDKP: Synthesis, Role in ACE Inhibition, and Its Therapeutic Potential in Hypertension and Cardiovascular Diseases.” Pharmacological Research, vol. 134, Aug. 2018, pp. 268–279, 10.1016/j.phrs.2018.07.006.
- Peng, Hongmei, et al. “Ac-SDKP Inhibits Transforming Growth Factor-β1-Induced Differentiation of Human Cardiac Fibroblasts into Myofibroblasts.” American Journal of Physiology-Heart and Circulatory Physiology, vol. 298, no. 5, May 2010, pp. H1357–H1364, 10.1152/ajpheart.00464.2009.
- Li, Shifeng, et al. “[Inhibition Effect of N-Acetyl-Seryl-Aspartyl-Lysyl-Proline on Myofibroblast Differentiation of MRC-5 Human Fetal Lung Fibroblasts Inuced by Ang II].” Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi = Zhonghua Laodong Weisheng Zhiyebing Zazhi = Chinese Journal of Industrial Hygiene and Occupational Diseases, vol. 32, no. 11, 1 Nov. 2014, pp. 801–805, pubmed.ncbi.nlm.nih.gov/25579022/.
- Xu, Hong, et al. “A New Antifibrotic Target of Ac-SDKP: Inhibition of Myofibroblast Differentiation in Rat Lung with Silicosis.” PLoS ONE, vol. 7, no. 7, 3 July 2012, p. e40301, 10.1371/journal.pone.0040301.
- Sharma, Umesh C., et al. “A Small Peptide Ac-SDKP Inhibits Radiation-Induced Cardiomyopathy.” Circulation: Heart Failure, vol. 11, no. 8, Aug. 2018, 10.1161/circheartfailure.117.004867.
- Cavasin, Maria A., et al. “Decreased Endogenous Levels of Ac-SDKP Promote Organ Fibrosis.” Hypertension, vol. 50, no. 1, July 2007, pp. 130–136, 10.1161/hypertensionaha.106.084103.
- Kassem, Kamal M., et al. “Tβ4–Ac-SDKP Pathway: Any Relevance for the Cardiovascular System?” Canadian Journal of Physiology and Pharmacology, vol. 97, no. 7, 1 July 2019, pp. 589–599, www.ncbi.nlm.nih.gov/pmc/articles/PMC6824425/, 10.1139/cjpp-2018-0570.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
RUO Disclaimer
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.
Why Choose Our Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment)?
When you are looking for Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) for sale, quality is paramount.
Our products undergo rigorous testing to ensure they meet the strict requirements of laboratory environments.
By choosing to buy Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) from our store, you are guaranteed a product that is:
- High Purity (Tested for 99%+)
- Fast Shipping – Always in stock
- Secure Packaging for Research Integrity
- Competitive Pricing for Bulk Orders
Specifications & Technical Data
| Feature | Specification |
|---|---|
| Product Name | Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) |
| SKU | 115 |
| Purity | >99% |
| Form | Research Grade Compound |
| Availability | In Stock / For Sale |
Scientific Research & Clinical Applications
The research surrounding Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) is vast. Scientists explore its potential in various metabolic and physiological models.
For more detailed scientific data, you can visit PubMed
to review the latest peer-reviewed literature regarding this compound.
Frequently Asked Questions
Where can I buy Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment)?
You can buy Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) directly from our website. We provide a secure checkout and fast shipping to ensure your research stays on track.
Is Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) in stock?
Yes, we currently have Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment) in stock. Orders are typically processed within 24 hours to ensure rapid delivery to your laboratory.
Related Research Products
If you are interested in Repair and Recovery (60 Capsules) (Stable BPC-157 Arginate, Thymosin Beta-4 Fragment), you may also want to explore these related products currently in stock:
Disclaimer: All products listed are for research purposes only. Not for human consumption.
Related products
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides











