LL-37 5mg (CAP-18)
$95.00
LL-37 is a cationic α-helical research peptide derived from the C-terminal region of human cathelicidin (hCAP18). It exhibits broad-spectrum antimicrobial and immunomodulatory activity in preclinical studies, acting via membrane disruption and modulation of TLR/NF-κB pathways. LL-37 is widely used in microbiology and immunology research investigating host defense peptides and innate immune signaling.
For research use only. Not for human consumption.
References:
Dürr UH et al., Biochim Biophys Acta, 2006 1758(9):1408–1425
Zanetti M et al., J Leukoc Biol, 2004 75(1):39–48
Vandamme D et al., Cell Mol Life Sci, 2012 69(2):210–227
LL-37 5mg (CAP-18) – Buy High-Quality LL-37 5mg (CAP-18) Online
Looking to buy LL-37 5mg (CAP-18) for your research laboratory? You have come to the right place.
We currently have LL-37 5mg (CAP-18) for sale and it is in stock and ready for immediate shipping.
Our LL-37 5mg (CAP-18) is of the highest purity, making it the best LL-37 5mg (CAP-18) online for scientific studies.
Product Overview
LL-37 5mg (CAP-18) is a premium research compound widely utilized in various scientific studies.
Researchers seeking to buy LL-37 5mg (CAP-18) online often prioritize purity and consistency.
This compound has been studied extensively for its unique biochemical properties and its role in cellular pathways.
LL-37: Overview
LL-37 (also referenced as CAP-18–derived peptide) is a cationic, amphipathic cathelicidin-family peptide extensively used as a research tool for interrogating innate immune signaling, microbe–membrane interactions, and host-cell modulatory responses. Cathelicidins are expressed by multiple leukocyte lineages and epithelial compartments and are frequently studied as endogenous effectors within barrier defense systems. In experimental systems, LL-37 is characterized as a pleiotropic mediator whose activity is highly dependent on local milieu (ionic strength, serum components, membrane composition) and cellular activation state, making it valuable for mechanistic dissection of context-dependent immunobiology.
In preclinical literature, LL-37 has been used to probe antimicrobial membrane disruption, lipopolysaccharide (LPS) binding/neutralization, chemotactic gradients, cytokine network modulation, epithelial restitution programs, and vascular signaling modules in vitro and in vivo (animal models). This product page is provided in a laboratory-focused format to support experimental design, assay selection, and interpretation of mechanistic readouts.
LL-37: Biochemical Characteristics

Source: PubChem
Sequence: -Leu-Leu-Gly-Asp-Phe-Phe-Arg-Lys-Ser-Lys-Glu-Lys-Ile-Gly-Lys-Glu-Phe-Lys-Arg-Ile-Val-Gln-Arg-Ile-Lys-Asp-Phe-Leu-Arg-Asn-Leu-Val-Pro-Arg-Thr-Glu-Ser
Molecular Formula: C205H340N60O53
Molecular Weight: 4493.342 g/mol
PubChem CID: 16198951
CAS Number: 154947-66-7
Synonyms: CAP-18, Cathelicidin, antibacterial peptide LL-37
LL-37 is a short, positively charged peptide with amphipathic character that supports interactions with anionic lipid surfaces and microbial envelope components. In biophysical studies, LL-37 can adopt conformations consistent with membrane-associated states, enabling pore formation and/or membrane destabilization under defined conditions. Experimental outcomes can vary with buffer composition, ionic strength, and the presence of serum proteins that sequester or inhibit peptide activity.
LL-37: Research Applications
LL-37 is commonly employed as a mechanistic probe across multiple preclinical research settings, including:
- Host–pathogen interface assays: evaluation of peptide–membrane interactions, microbial growth inhibition, and synergy studies with other innate defense factors (e.g., defensins, lysozyme) in controlled in vitro systems.
- Pattern-recognition and innate signaling studies: interrogation of LPS binding/neutralization and downstream modulation of innate immune pathways in cell-based assays.
- Leukocyte behavior assays: chemotaxis and migration studies in neutrophils/eosinophils and immune-cell activation-state–dependent response profiling.
- Epithelial barrier models: scratch/wound-closure assays, epithelial migration, and barrier integrity measurements under inflammatory stimuli in culture models.
- Vascular biology models: endothelial signaling experiments focused on eicosanoid pathways and angiogenic programs in vitro and in vivo (animal models).
- Inflammation-network mapping: cytokine and interferon response profiling (e.g., IFN-α, IL-18) and macrophage polarization response modulation in vitro.
Selection of assay format and controls should account for known matrix effects (e.g., serum inhibition), peptide adsorption to plastics, and concentration-dependent cytotoxicity observed in some experimental contexts reported in preclinical literature.
LL-37: Pathway / Mechanistic Context
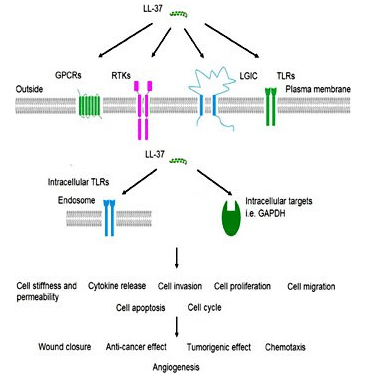
Source: Karger
Mechanistically, LL-37 is frequently studied in the context of:
- Microbial envelope targeting: interaction with LPS and other anionic surface structures, leading to membrane perturbation and loss of envelope integrity in susceptible organisms.
- Innate immune receptor cross-talk: modulation of pattern-recognition receptor signaling, including reported effects involving Toll-like receptor pathways in cell-based and animal-model studies.
- Chemotactic and immunomodulatory signaling: regulation of leukocyte trafficking signals and context-dependent adjustment of cytokine outputs that vary with cell type and activation state.
- Epithelial restitution programs: engagement of migratory/proliferative responses in epithelial models, used to map barrier restoration signaling under experimental injury paradigms.
- Eicosanoid-mediated vascular signaling: induction of prostaglandin E2 (PGE2) signaling modules in endothelial systems, including EP receptor–linked angiogenic programs reported in animal and cell models.
Because LL-37 can function as both a direct membrane-active peptide and a host-cell response modulator, experimental interpretation benefits from orthogonal readouts (membrane integrity assays, receptor/pathway reporter assays, transcript/protein cytokine profiling, and viability controls) to differentiate primary membrane effects from downstream signaling events.
LL-37: Preclinical Research Summary
Inflammation-Associated Models
In preclinical and ex vivo systems, LL-37 is repeatedly described as a context-dependent immunomodulator. Reported observations include altered keratinocyte survival programs, interferon and interleukin production changes (including IFN-α and IL-18 in defined models), chemotaxis effects in granulocyte lineages, and modulation of receptor-linked inflammatory signaling networks (including reported effects involving TLR4-associated signaling in selected assay systems)[1].
Cell-based studies also indicate that immune-cell activation state can shift LL-37–linked outcomes, including differential inflammatory signatures in lymphocyte populations under distinct stimulation conditions[2]. These findings support the use of LL-37 as a tool for mapping homeostatic versus pro-inflammatory network behaviors in tightly controlled experimental designs.
Antimicrobial and LPS-Interaction Studies
LL-37 is widely used in antimicrobial mechanism-of-action experiments, including studies of inducible peptide accumulation in barrier tissues under infectious challenge and synergy experiments with other innate defense factors such as human beta-defensin 2[3]. A common mechanistic emphasis is LL-37 binding to LPS, with experimental readouts examining LPS-neutralization capacity, antimicrobial potency, and matrix-dependent inhibition (e.g., serum-associated attenuation) in vitro[4].
Although membrane-targeting activity is frequently highlighted in Gram-negative contexts, LL-37 has also been studied against Gram-positive organisms. In vitro reports describe cooperative effects with lysozyme and defensins against organisms such as Staphylococcus aureus, supporting its use as a probe for envelope-stress and multi-effector synergy paradigms[5].
Airway and Epithelial Model Systems
Airway-focused preclinical studies examine LL-37 in the setting of inhaled inflammatory triggers and barrier perturbation models, often using LPS-driven stimuli to interrogate innate response dynamics. In epithelial culture models, LL-37 has been reported to stimulate airway epithelial proliferation and scratch-wound closure responses, enabling pathway interrogation of epithelial restitution and repair-associated signaling in vitro[7].
Arthritis-Linked Experimental Literature
In rodent studies and observational analyses in inflamed joint environments, cathelicidins (including LL-37 and related species orthologs) have been evaluated for associations with inflammatory events and tissue remodeling signatures[8]. Separate work in mouse models reports no evidence for a required pathogenic role of cathelicidins in select lupus/arthritis model settings, supporting the interpretation that cathelicidin upregulation can reflect inflammatory-state biomarkers rather than necessary drivers in some paradigms[9].
Derivatives of LL-37 have also been used as mechanistic tools in collagen-induced arthritis models to interrogate collagen damage–associated pathways and antibody-response correlates in vivo (murine), including modulation of readouts linked to type II collagen reactivity[10]. Additional in vitro work evaluates LL-37/derivatives within cytokine-driven inflammation models (e.g., IL-32–stimulated systems) to map upstream/downstream signaling responses[11].
Studies also evaluate Toll-like receptor expression dynamics in synovial fibroblast systems (e.g., TLR3 upregulation under immune-cell influence) as a framework for assessing how LL-37 may intersect with PRR-linked inflammatory amplification circuits in vitro and in animal models[12]. Macrophage-focused work further reports selective attenuation of pro-inflammatory macrophage responses under defined conditions, emphasizing the peptide’s use for dissecting polarization-associated signaling outputs[13].
Intestinal Barrier Models and Related Systems
In vitro intestinal epithelial systems report LL-37–linked effects on epithelial migration and barrier integrity under inflammatory stress conditions, supporting its frequent use in barrier restitution assays and apoptosis/viability profiling within controlled stimuli frameworks[14]. Additional cell-culture studies describe cooperative behavior between beta-defensin 2 and epithelial repair processes, including reduced TNF-related cell death markers under defined experimental conditions[15].
Cancer-Adjacent Preclinical Mechanisms
Preclinical literature describes mixed, context-dependent observations regarding LL-37 in tumor biology models. Mechanistic discussions frequently focus on tumor microenvironment immunobiology, including macrophage-linked programs and vitamin D–associated regulation of antimicrobial peptide expression in experimental systems[16].
Endothelial Signaling and Angiogenesis Models
LL-37 has been reported to induce endothelial PGE2 signaling and EP receptor–linked angiogenic responses in vitro and in vivo (animal model), offering a tractable framework for studying eicosanoid-driven vascular remodeling modules under defined experimental conditions[17].
LL-37: Form & Analytical Testing
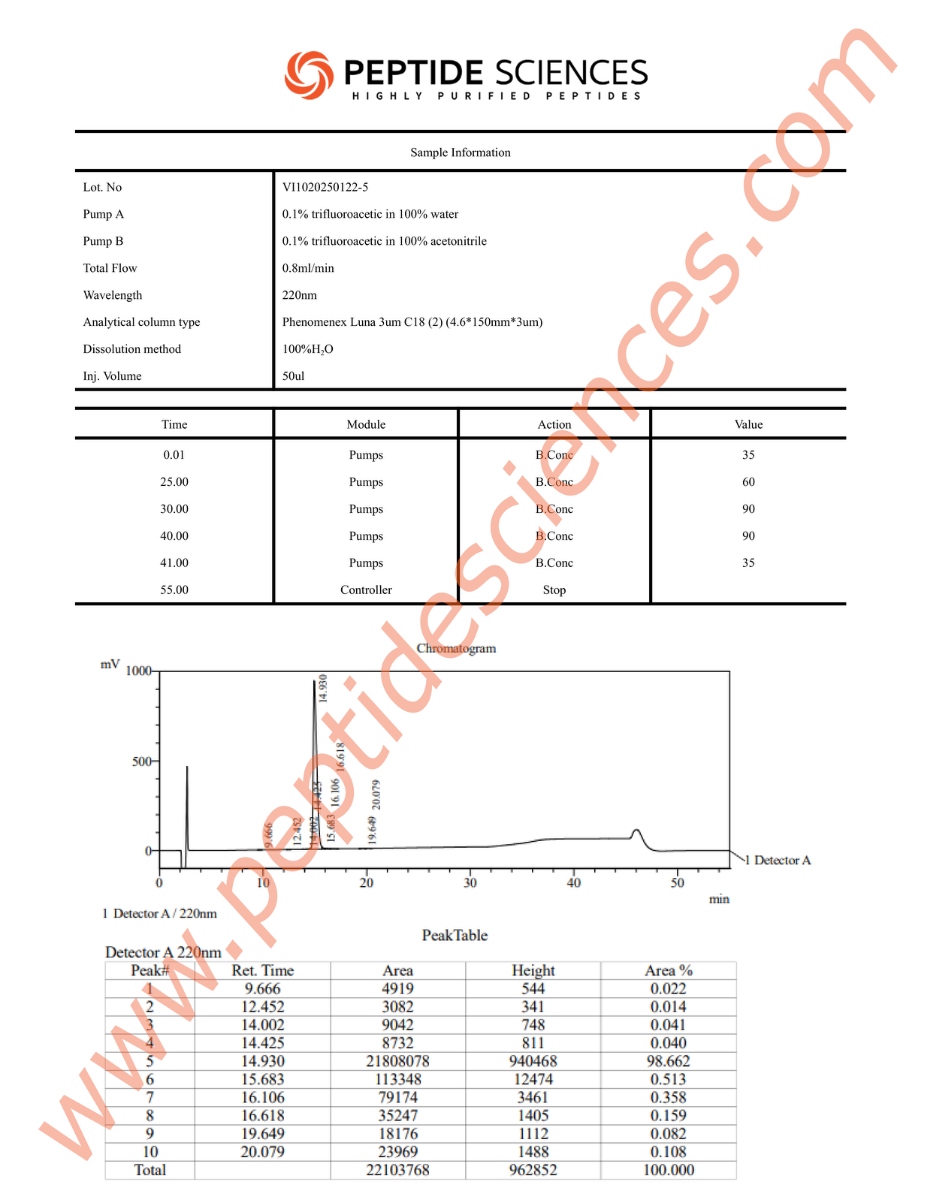
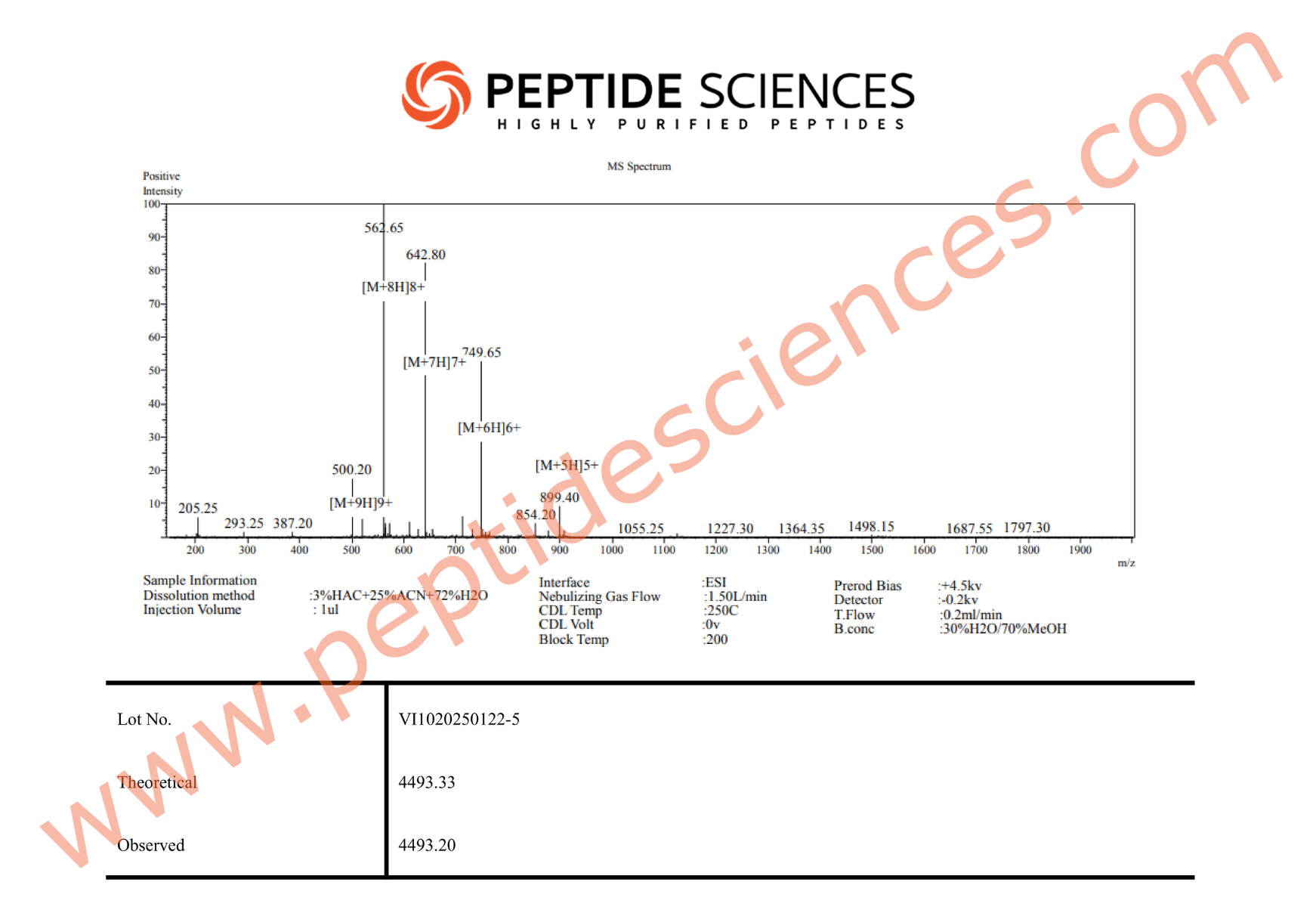
This RUO peptide is supplied for laboratory experimentation. Standard analytical characterization for synthetic peptides commonly includes orthogonal identity and purity confirmation approaches (e.g., mass spectrometry and chromatographic methods). Researchers should implement handling procedures appropriate for peptides, including minimizing repeated freeze–thaw cycles, reducing adsorption losses to plastics, and selecting solvent/buffer systems compatible with downstream assays.
Because LL-37 activity is sensitive to assay matrix conditions, experimental controls are recommended to account for serum binding/inhibition, ionic strength effects, and concentration-dependent cytotoxicity observed in some in vitro formats reported in the literature.
For structure–function studies, LL-37’s distinct conformational behavior across species and sequence variants provides a useful platform for probing how amino-acid substitutions alter membrane association, receptor engagement, and downstream signaling outputs in controlled experimental models[18].
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Daniela Xhindoli, PhD. operates from the University of Trieste, UNITS · Department of Life Sciences. Her focus surrounds gram-negative bacteria, the biological activities of LL-37 on simultaneously modulating pro-inflammatory and anti-inflationary pathways, and the antibacterial and antimicrobial effects of LL-37.
Daniela Xhindoli, PhD. is being referenced as one of the leading scientists involved in the research and development of LL-37. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Daniela Xhindoli, PhD. is listed in [18] under the referenced citations.
Referenced Citations
- J. M. Kahlenberg and M. J. Kaplan, “Little peptide, big effects: the role of LL-37 in inflammation and autoimmune disease,” J. Immunol. Baltim. Md 1950, vol. 191, no. 10, Nov. 2013.
- D. S. Alexandre-Ramos et al., “LL-37 treatment on human peripheral blood mononuclear cells modulates immune response and promotes regulatory T-cells generation,” Biomed. Pharmacother. Biomedecine Pharmacother., vol. 108, pp. 1584–1590, Dec. 2018.
- P. Y. Ong et al., “Endogenous antimicrobial peptides and skin infections in atopic dermatitis,” N. Engl. J. Med., vol. 347, no. 15, pp. 1151–1160, Oct. 2002.
- C. D. Ciornei, T. Sigurdardóttir, A. Schmidtchen, and M. Bodelsson, “Antimicrobial and chemoattractant activity, lipopolysaccharide neutralization, cytotoxicity, and inhibition by serum of analogs of human cathelicidin LL-37,” Antimicrob. Agents Chemother., vol. 49, no. 7, pp. 2845–2850, Jul. 2005.
- X. Chen et al., “Synergistic effect of antibacterial agents human β-defensins, cathelicidin LL-37 and lysozyme against Staphylococcus aureus and Escherichia coli,” J. Dermatol. Sci., vol. 40, no. 2, pp. 123–132, Nov. 2005.
- M. Golec, “Cathelicidin LL-37: LPS-neutralizing, pleiotropic peptide,” Ann. Agric. Environ. Med. AAEM, vol. 14, no. 1, pp. 1–4, 2007.
- R. Shaykhiev et al., “Human endogenous antibiotic LL-37 stimulates airway epithelial cell proliferation and wound closure,” Am. J. Physiol. Lung Cell. Mol. Physiol., vol. 789, no. 5, pp. L842-848, Nov. 2005.
- M. H. Hoffmann et al., “The cathelicidins LL-37 and rCRAMP are associated with pathogenic events of arthritis in humans and rats,” Ann. Rheum. Dis., vol. 72, no. 7, pp. 1239–1248, Jul. 2013.
- D. Kienhöfer et al., “No evidence of pathogenic involvement of cathelicidins in patient cohorts and mouse models of lupus and arthritis,” PloS One, vol. 9, no. 12, p. e115474, 2014.
- L. N. Y. Chow et al., “Human cathelicidin LL-37-derived peptide IG-19 confers protection in a murine model of collagen-induced arthritis,” Mol. Immunol., vol. 57, no. 2, pp. 86–92, Feb. 2014.
- K.-Y. G. Choi, S. Napper, and N. Mookherjee, “Human cathelicidin LL-37 and its derivative IG-19 regulate interleukin-32-induced inflammation,” Immunology, vol. 143, no. 1, pp. 68–80, Sep. 2014.
- W. Zhu et al., “Arthritis is associated with T-cell-induced upregulation of Toll-like receptor 3 on synovial fibroblasts,” Arthritis Res. Ther., vol. 13, no. 3, p. R103, Jun. 2011.
- K. L. Brown et al., “Host defense peptide LL-37 selectively reduces proinflammatory macrophage responses,” J. Immunol. Baltim. Md 1950, vol. 186, no. 9, pp. 5497–5505, May 2011.
- J.-M. Otte et al., “Effects of the cathelicidin LL-37 on intestinal epithelial barrier integrity,” Regul. Pept., vol. 156, no. 1–3, pp. 104–117, Aug. 2009.
- J.-M. Otte et al., “Human beta defensin 2 promotes intestinal wound healing in vitro,” J. Cell. Biochem., vol. 104, no. 6, pp. 2286–2297, Aug. 2008.
- X. Chen et al., “Roles and Mechanisms of Human Cathelicidin LL-37 in Cancer,” Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol., vol. 47, no. 3, pp. 1060–1073, 2018.
- Salvado M. Dolores, Di Gennaro Antonio, Lindbom Lennart, Agerberth Birgitta, and Haeggström Jesper Z., “Cathelicidin LL-37 Induces Angiogenesis via PGE2–EP3 Signaling in Endothelial Cells, In Vivo Inhibition by Aspirin,” Arterioscler. Thromb. Vasc. Biol., vol. 33, no. 8, pp. 1965–1972, Aug. 2013.
- D. Xhindoli, S. Pacor, M. Benincasa, M. Scocchi, R. Gennaro, and A. Tossi, “The human cathelicidin LL-37 — A pore-forming antibacterial peptide and host-cell modulator,” Biochim. Biophys. Acta BBA – Biomembr., vol. 1858, no. 3, pp. 546–566, Mar. 2016.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
RUO Disclaimer
For Laboratory Research Only. Not for human use, medical use, diagnostic use, or veterinary use.


Storage Instructions:
All of our products are manufactured using the Lyophilization (Freeze Drying) process, which ensures that our products remain 100% stable for shipping for up to 3-4 months.
Once the peptides are reconstituted (mixed with bacteriostatic water), they must be stored in the fridge to maintain stability. After reconstitution, the peptides will remain stable for up to 30 days.
Lyophilization is a unique dehydration process, also known as cryodesiccation, where the peptides are frozen and then subjected to low pressure. This causes the water in the peptide vial to sublimate directly from solid to gas, leaving behind a stable, crystalline white structure known as lyophilized peptide. The puffy white powder can be stored at room temperature until you’re ready to reconstitute it with bacteriostatic water.
Once peptides have been received, it is imperative that they are kept cold and away from light. If the peptides will be used immediately, or in the next several days, weeks or months, short-term refrigeration under 4C (39F) is generally acceptable. Lyophilized peptides are usually stable at room temperatures for several weeks or more, so if they will be utilized within weeks or months such storage is typically adequate.
However, for longer term storage (several months to years) it is more preferable to store peptides in a freezer at -80C (-112F). When storing peptides for months or even years, freezing is optimal in order to preserve the peptide’s stability.
Why Choose Our LL-37 5mg (CAP-18)?
When you are looking for LL-37 5mg (CAP-18) for sale, quality is paramount.
Our products undergo rigorous testing to ensure they meet the strict requirements of laboratory environments.
By choosing to buy LL-37 5mg (CAP-18) from our store, you are guaranteed a product that is:
- High Purity (Tested for 99%+)
- Fast Shipping – Always in stock
- Secure Packaging for Research Integrity
- Competitive Pricing for Bulk Orders
Specifications & Technical Data
| Feature | Specification |
|---|---|
| Product Name | LL-37 5mg (CAP-18) |
| SKU | 77 |
| Purity | >99% |
| Form | Research Grade Compound |
| Availability | In Stock / For Sale |
Scientific Research & Clinical Applications
The research surrounding LL-37 5mg (CAP-18) is vast. Scientists explore its potential in various metabolic and physiological models.
For more detailed scientific data, you can visit PubMed
to review the latest peer-reviewed literature regarding this compound.
Frequently Asked Questions
Where can I buy LL-37 5mg (CAP-18)?
You can buy LL-37 5mg (CAP-18) directly from our website. We provide a secure checkout and fast shipping to ensure your research stays on track.
Is LL-37 5mg (CAP-18) in stock?
Yes, we currently have LL-37 5mg (CAP-18) in stock. Orders are typically processed within 24 hours to ensure rapid delivery to your laboratory.
Related Research Products
If you are interested in LL-37 5mg (CAP-18), you may also want to explore these related products currently in stock:
Disclaimer: All products listed are for research purposes only. Not for human consumption.
Related products
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides
All Peptides